A cluster of COVID-19 in Beijing, People’s Republic of China was reported
On Jun. 13, 2020, officials from the WHO National Health Commission and Beijing Health Commission announced preliminary investigations…
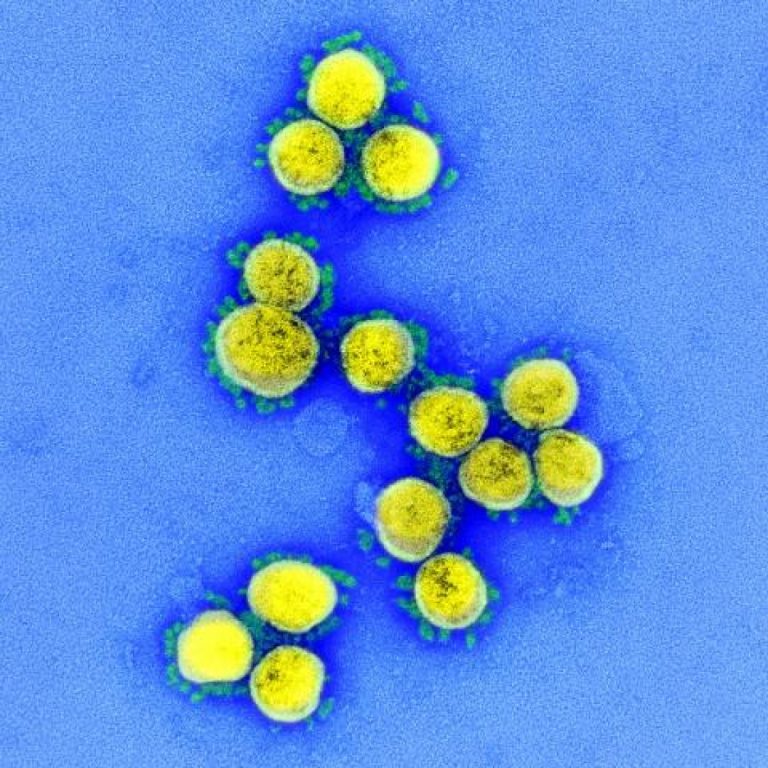
On Jun. 13, 2020, officials from the WHO National Health Commission and Beijing Health Commission announced preliminary investigations…

On Jun. 13, 2020, Sinovac Biotech announced positive preliminary results of phase I/II clinical trial for the Companyメs…
On Jun. 13, 2020, Dr. Reddy’s Laboratories announced that it had entered into a non-exclusive Licensing Agreement with…

On Jun. 13, 2020, AstraZeneca announced an agreement with Europeメs Inclusive Vaccines Alliance (IVA), spearheaded by Germany, France,…

On Jun. 12, 2020, a tiny genetic mutation in the SARS coronavirus 2 variant circulating throughout Europe and…

On Jun. 12, 2020, the FDA announced it had approved an expanded indication for Merck’s GARDASIL9 for the…

On Jun. 12, 2020, the California Institute for Regenerative Medicine (CIRM) awarded $349,999 to Dr. Vaithilingaraja Arumugaswami at…
On Jun. 12, 2020, Cue Health announced that it had received Emergency Use Authorization from the U.S. Food…
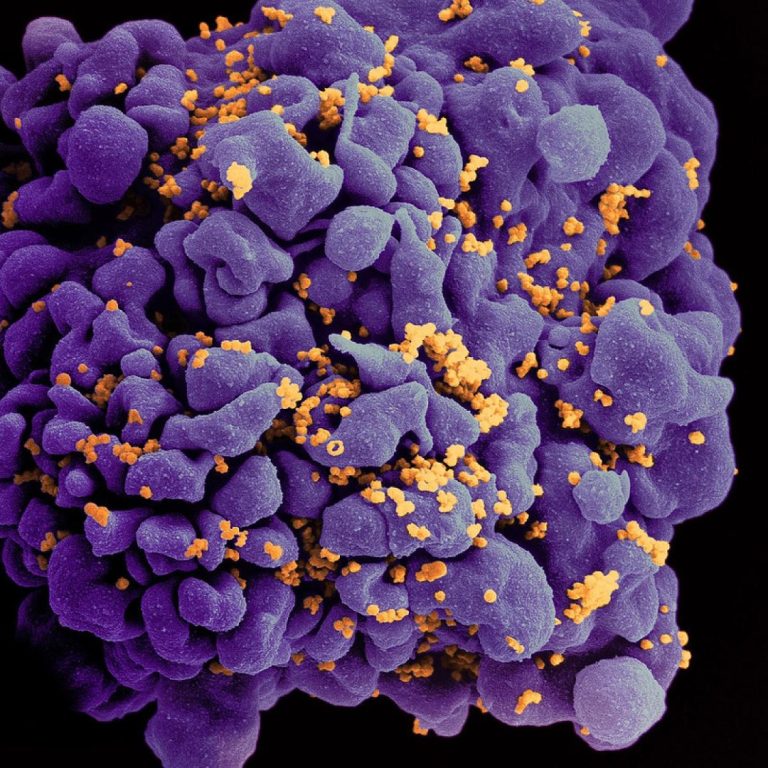
On Jun. 12, 2020, the U.S. Food and Drug Administration (FDA) approved ViiV Healthcare’s Tivicay (dolutegravir) tablets and…

On Jun. 12, 2020, ViiV Healthcare, a GSK company, with Pfizer and Shionogi Limited, announced the FDA had…

On Jun. 11, 2020, CHF Solutions announced that it has sold Aquadex SmartFlowル consoles and disposables to a…

On Jun. 11, 2020, Atossa Therapeutics announced that the manuscript of the results from in vitro testing of…

On Jun. 11, 2020, InBios announced that it had received Emergency Use Authorization (EUA) from the U.S. Food…

On Jun. 11, 2020, Regeneron announced initiation of the first clinical trial of REGN-COV2, its investigational dual antibody…
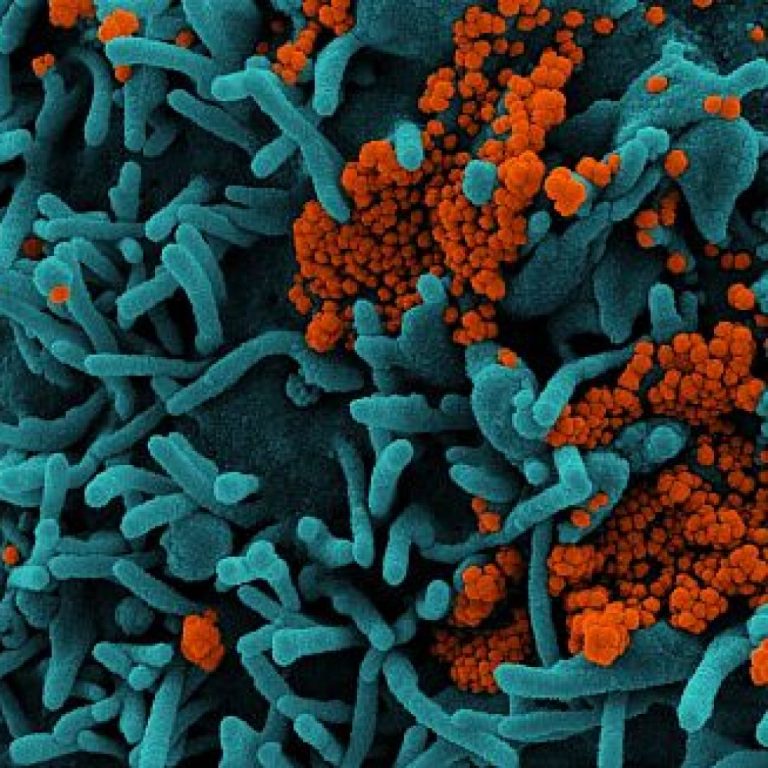
On Jun. 11, 2020, Regeneron announced that Science had accepted for publication two papers describing the creation of…

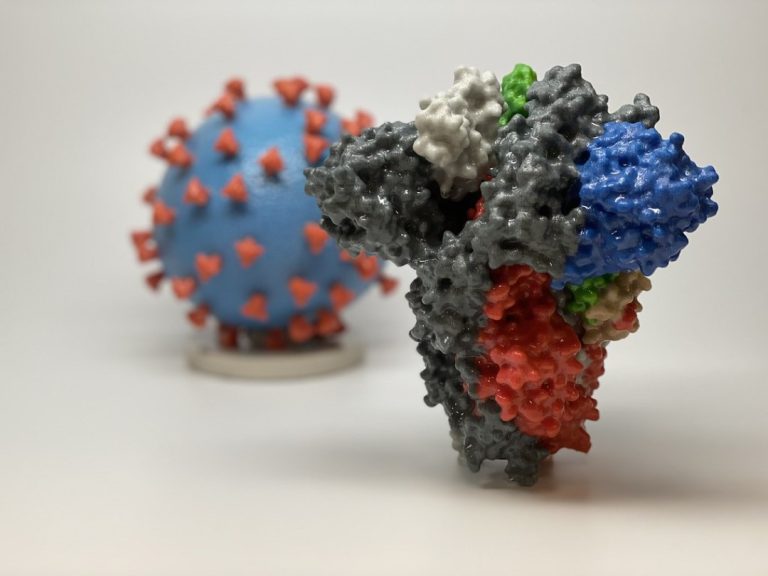
On Jun. 11, 2020, a new study led by UT Southwestern researchers revealed the structure of the active…

On Jun. 11, 2020, BioCryst Pharmaceuticals announced new data published in Science Translational Medicine that show, in a…

On Jun. 11, 2020, a team of researchers from the National Library of Medicine identified genomic features of…

On Jun. 11, 2020, Bristol-Myers Squibb announced that Opdivo (nivolumab) was approved by the FDA for the treatment…

On Jun. 11, 2020, scientists announced the development of a new test that can help identify people who…
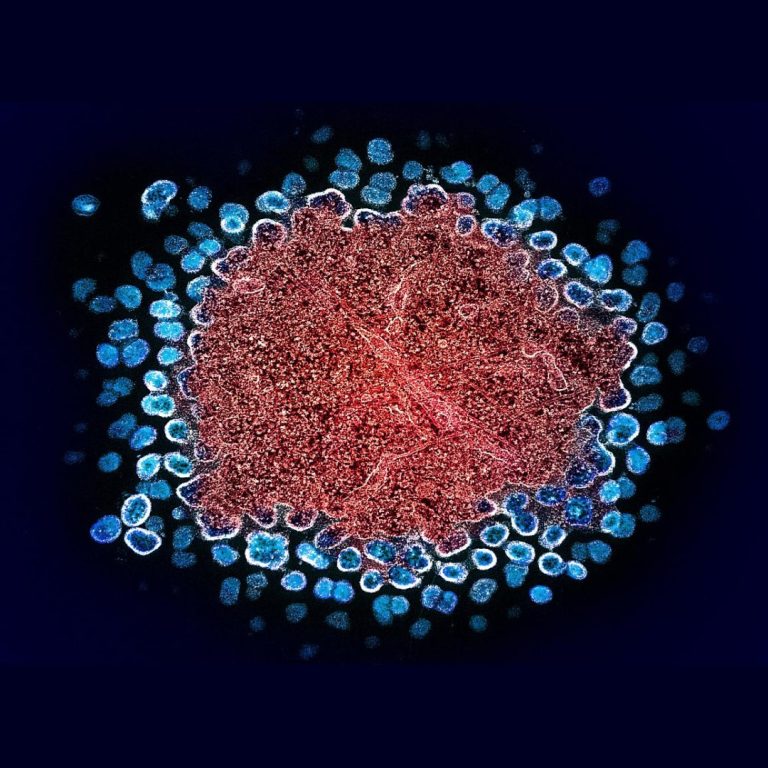
On Jun. 11, 2020, researchers have found that astrocytes, a type of brain cell can harbor HIV and…

On Jun. 11, 2020, the FDA approved Viela Bio’s Uplizna (inebilizumab-cdon) injection for intravenous use for the treatment…
On Jun. 11, 2020, the World Trade Organizationメs (WTO) reported that it was following up with Chinese authorities…

On Jun. 11, 2020, scientists at the Universities of Oxford, Exeter, Yunnan and Bristol announced they had discovered…

On Jun. 11, 2020, Sinovac Biotech announced the signing of a clinical development collaboration agreement to advance the…

On Jun. 11, 2020, Emergent BioSolutions announced deployment of its molecule-to-market contract development and manufacturing (CDMO) services to…

On Jun. 11, 2020, Quidel announced it had received funding from the Biomedical Advanced Research and Development Authority…

On Jun. 11, 2020, AIM ImmunoTech announced that it had filed a provisional patent application for, among other…

On Jun. 11, 2020, Moderna announced progress on late-stage development of mRNA-1273, the Company’s mRNA vaccine candidate against…

On Jun. 11, 2020, Innovation Pharma announced the Company and researchers at a U.S. Regional Biocontainment Laboratory (RBL)…