Calquence showed promising clinical improvement in majority of 19 hospitalised COVID-19 patients
On Jun. 5, 2020, results were published in Science Immunology showed that Calquence (acalabrutinib), a Brutonメs tyrosine kinase…

On Jun. 5, 2020, results were published in Science Immunology showed that Calquence (acalabrutinib), a Brutonメs tyrosine kinase…
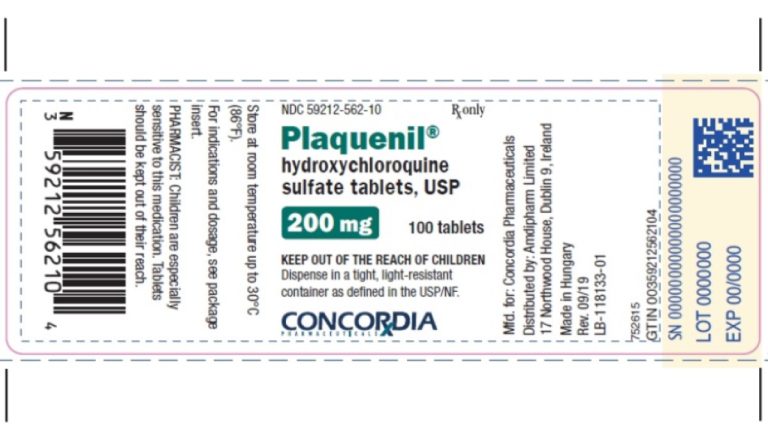
On Jun. 5, 2020, the Chief Investigators of the randomised evaluation of COVid-19 thERapY (RECOVERY) trial on hydroxychloroquine…

On Jun. 5, 2020, BioSig Technologies and its subsidiary, ViralClear Pharma, announced it had expanded its patient enrollment…

On Jun. 5, 2020, the UK thelped secure $7.4 billion in funding to support global vaccine supply and…

On Jun. 5, 2020, CEPI, the Coalition for Epidemic Preparedness Innovations, CSL and The University of Queensland (UQ)…

On Jun. 4, 2020, the Bill & Melinda Gates Foundation announced a five-year, US$1.6 billion commitment to Gavi,…

On Jun. 4, 2020, AstraZeneca announced the next steps in its commitment to broad and equitable global access…

On Jun. 4, 2020, world leaders pledged an additional US$ 8.8 billion for Gavi, the Vaccine Alliance, far…

On Jun. 4, 2020, vaccine manufacturers MSD, GSK, Innovax, Serum Institute of India and Walvax pledged to ramp…

On Jun. 4, 2020, the U.S. Department of Health and Human Services (HHS) announced new Guidance that specified…

On Jun. 4, 2020, vaccine manufacturers MSD, GSK, Innovax, Serum Institute of India (SII) and Walvax pledged to…
On Jun. 4, 2020, the World Health Organization (WHO) welcomed funding commitments from Gavi, the Vaccine Alliance’s third…

On Jun. 4, 2020, the WHO announced new pledges that will enable Gavi to protect the next generation…
On Jun. 4, 2020, the FDA approved Merck’s Recarbrio (a combination of imipenem-cilastatin and relebactam) to treat hospital-acquired…

On Jun. 4, 2020, data from the Broad Institute of MIT and Harvard, Massachusetts General Hospital, and the…

On Jun. 4, 2020, Novavax announced that the company had been awarded a contract by the U.S. Department…

On Jun. 4, 2020, Agricultural Research Service scientists and their Oregon State University collaborators announced they had developed…

On Jun. 4, 2020, INOVIO and Seoul National University Hospital announced a partnership to start a Phase 1/2…

On Jun. 4, 2020, iBio announced it had initiatied preclinical immunization studies for its second COVID-19 vaccine platform….

On Jun. 4, 2020, with funding from the Fralin Life Sciences Institute at Virginia Tech, researchers James Weger-Lucarelli…

On Jun. 4, 2020, Roche announced the initiation of a global phase III, randomised, double-blind, multicentre study (REMDACTA)…

On Jun. 3, 2020, in a nationwide study, NIH funded researchers found that the presence of abnormal bundles…

On Jun. 3, 2020, CEPI, the Coalition for Epidemic Preparedness Innovations, in collaboration with Ind-CEPI, announced a new…

On Jun. 2, 2020, Agenus announced the FDA’s clearance of AgenTus’ IND application for an allogeneic iNKT therapy….

On Jun. 2, 2020, Pfizer announced the establishment of the Pfizer Breakthrough Growth Initiative, through which Pfizer will…

On Jun. 2, 2020, the USDA announced the first confirmed case of SARS-CoV-2 (the virus that causes COVID-19)…

On Jun. 2, 2020, Albert Einstein College of Medicine announced it had received a $1 million challenge grant…

On Jun. 2, 2020, Trevena announced it had entered into a collaboration with Imperial College London to evaluate…

On Jun. 2, 2020, the NIH reported that early results showed that remdesivir benefited hospitalized patients with severe…

On Jun. 1, 2020, the FDA issued an emergency use authorization (EUA) for Impella RP to include patients…