Revvity Secures FDA Approval for Improved Automated Latent Tuberculosis Test
On Apr. 2, 2025, Revvity announced that the U.S. Food and Drug Administration (FDA) has approved the Auto-Pure…
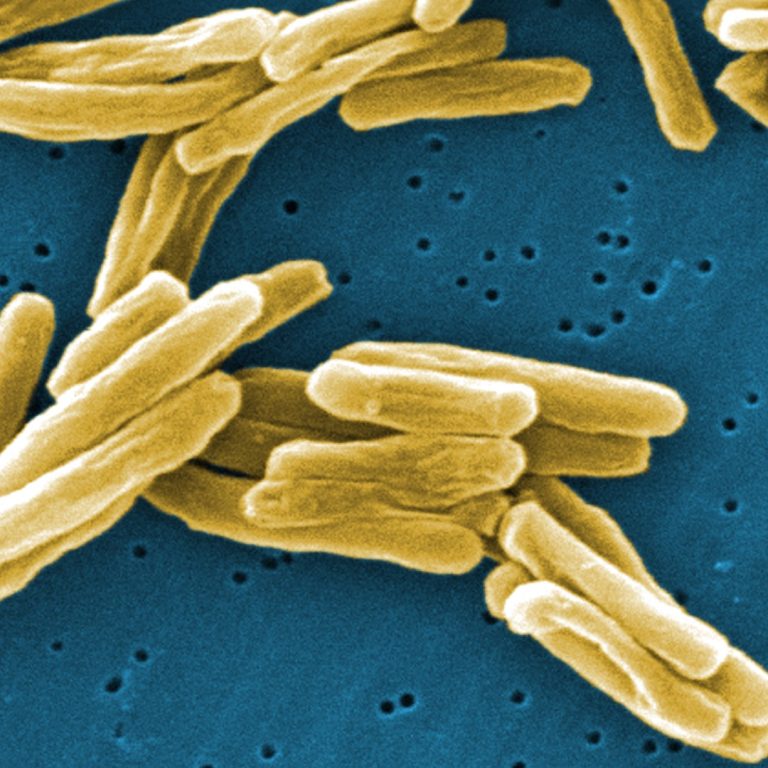
On Apr. 2, 2025, Revvity announced that the U.S. Food and Drug Administration (FDA) has approved the Auto-Pure…
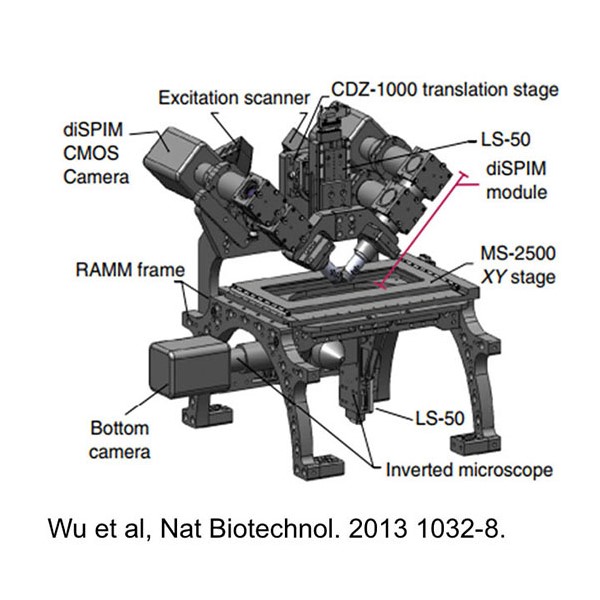
On Feb. 21, 2025, a team of researchers from the Marine Biological Laboratory (MBL) announced a hybrid custom-designed…

On Feb. 18, 2025, Hologic announced that the U.S. Food and Drug Administration (FDA) has granted 510(k) clearance…

On Feb. 11, 2025, Novartis announced an agreement to acquire Anthos Therapeutics, a Boston-based, privately held, clinical-stage biopharmaceutical…

On Feb. 5, 2025, scientists from the Cancer Dependency Map (DepMap) at the Broad Institute of MIT and…

On Jan. 30, 2025, the U.S. Food and Drug Administration (FDA) approved Vertex Pharmaceuticals’s Journavx (suzetrigine) 50 milligram…

On Jan. 27, 2025, researchers at the Broad Institute of MIT and Harvard, along with collaborators at Calico…
On Jan. 22, 2025, researchers at Dana-Farber Cancer Institute announced they have developed a breakthrough method to detect…

On Dec. 11, 2024, Massachusetts General Hospital (MGH), founding member of the Mass General Brigham healthcare system, announced…

On Oct. 15, 2024, Massachusetts Governor Maura Healey and the Massachusetts Life Sciences Center (MLSC) announced $21.4 million…
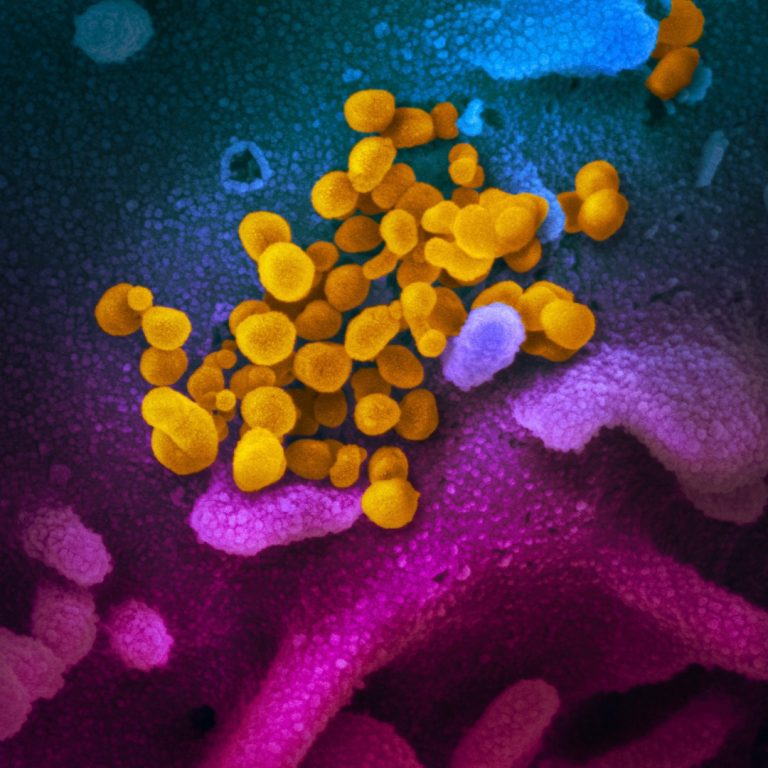
On Oct. 9, 2024, researchers from Brigham and Women’s Hospital found that people with wide-ranging long COVID symptoms…

On Oct. 7, 2024, The Nobel Foundation announced that Victor Ambros from Harvard University and Gary Ruvkun from…

On Sept. 25, 2024, a study led by scientists at Harvard Medical School announced they had developed an…
On Aug. 14, 2024, a study co-led by Mass General Brigham found that one in four patients with…

On May 31, 2024, Moderna announced that the U.S. Food and Drug Administration (FDA) had approved mRESVIA (mRNA-1345),…
On May 10, 2024, the National Academies of Sciences, Engineering, and Medicine reported that the COVID-19 vaccines made…

On Apr. 22, 2024, Moderna announced a contract with the Ministry of Health in Brazil (Ministerio da Saude)…

On Sept. 11, 2023, Moderna announced the U.S. Food and Drug Administration (FDA) had approved the supplemental Biologics…

On Sept. 6, 2023, Moderna announced that clinical trial data from its research assay confirmed its updated COVID-19…

On Aug. 27, 2003, The Institute for Collaborative Biotechnologies (ICB) announced it was funding a $50 million five…
On Aug. 25, 2023, Harvardメs Center for Brain Science announced the ceation of an eye atlas that pinpoints…

On Jun. 22, 2023 AquaBounty Technologies announced that the Company will pause the construction of its farm in…

On Apr. 20, 2023, Massachusetts Institute of Technology (MIT) researchers announced that have developed machine-learning algorithms that can…
On Apr. 4, 2023, National Institutes of Health (NIH) funded research was announced from the University of Rochester…

On Feb. 24, 2023, Moderna announced will make certain contingent development, commercial and regulatory milestone payments to the…

On Feb. 15, 2023, Moderna announced it will continue to offer its COVID-19 vaccines for free, even after…

On Dec. 21, 2022, Moderna announced the the finalization of a strategic partnership with the United Kingdom (UK)…

On Dec. 8, 2022, Moderna announced it had received emergency use authorization from the U.S. Food and Drug…

On Oct. 31, 2022, Moderna announced that it had received approval from the Ministry of Health, Labour and…

On Oct. 19, 2022, the U.S. Food and Drug Administration (FDA) recommended the use of the Novavax COVID-19…