Medicines and Healthcare Products Regulatory Agency authorized Moderna’s Omicron-containing bivalent booster in the UK
On Aug. 15, 2022, Moderna announced that the Medicines and Healthcare products Regulatory Agency (MHRA) in the UK…

On Aug. 15, 2022, Moderna announced that the Medicines and Healthcare products Regulatory Agency (MHRA) in the UK…

On Aug. 9, 2022, Moderna announced an amendment to its agreement with the European Commission (EC) to convert…

On Jul. 29, 2022, the U.S. Department of Health and Human Services (HHS), in collaboration with the U.S….

On Jul. 22, 2022, Moderna announced that the European Medicines Agency’s (EMA) Committee for Medicinal Products for Human…
On Jul. 18, 2022, Moderna announced that the Therapeutic Goods Administration (TGA) in Australia had granted provisional registration…

On Jul. 14, 2022, Moderna announced that Health Canada had approved the use of Moderna’s mRNA COVID-19 vaccine,…
On Jul. 11, 2022, the National Institute of Allergy and Infectious Diseases (NIAID) launched an early-stage clinical trial…

On Jun. 17, 2022, Moderna announced that it had received emergency use authorization from the U.S. Food and…

On Jun. 8, 2022, Moderna announced new clinical data on its Omicron-containing bivalent COVID booster candidate, mRNA-1273.214, containing…

On Jun. 2, 2022, Moderna announced an agreement with the European Commission (EC) to amend their originally agreed…

On May 31, 2022, Moderna and Takeda announced the transfer of the marketing authorization for Moderna’s COVID-19 vaccine…

On May 24, 2022, scientists at the Broad Institute of MIT and Harvard and the University of Massachusetts…

On May 19, 2022, the U.S. National Institutes of Health (NIH) announced that people who reported in a…
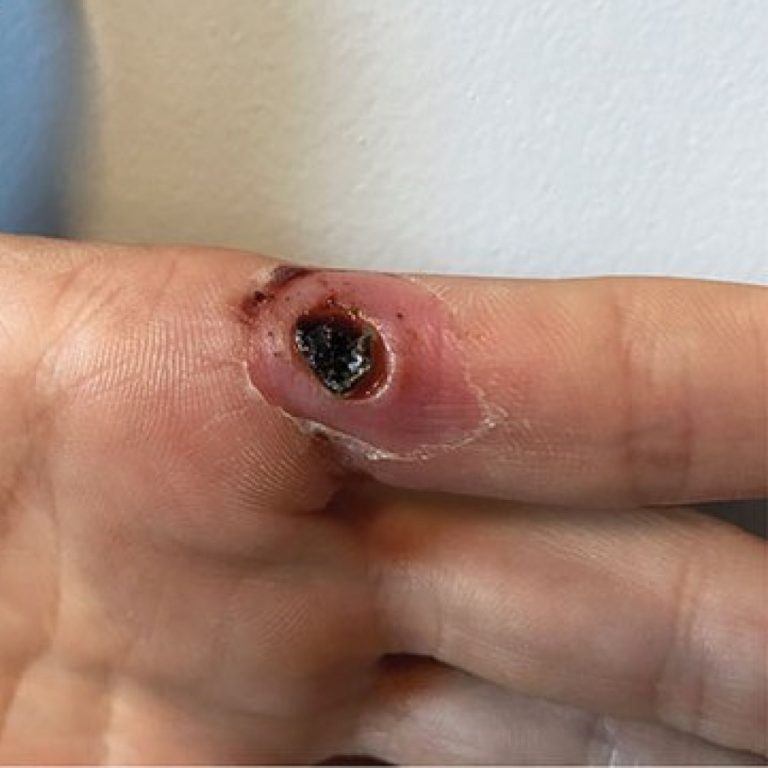
On May 18, 2022, scientists at the Centers for Disease Control and Prevention (CDC) announced that they were…

On Feb. 28, 2022, the Patent and Trial Appeal Board of the U.S. Patent and Trademark Office (USPTO)…

On Feb. 24, 2022, Moderna announced that the European Medicines Agency’s Committee for Medicinal Products for Human Use…

On Feb. 22, 2022, Moderna announced a distribution service agreement with Adium Pharma, a leading private Latin American…

On Feb. 16, 2022, Moderna announced that the Therapeutic Goods Administration in Australia had granted provisional registration for…

On Feb. 8, 2022, Moderna announced a new supply agreement with the government of Colombia for 10.8 million…

On Feb. 4, 2022, the Advisory Committee on Immunization Practices (ACIP) issued a standard recommendation for use of…

On Jan. 31, 2022, Moderna announced the U.S. Food and Drug Administration (FDA) had approved the Biologics License…

On Jan. 26, 2022, Moderna announced that the first participant had been dosed in the Phase 2 study…

On Dec. 27, 2021, Moderna announced that the Swiss Federal Government had exercised its option to purchase an…

On Dec. 27, 2021, Moderna announced a revised supply agreement with the government of South Korea for 20…
On Dec. 20, 2021, Moderna announced preliminary neutralizing antibody data against the Omicron variant following the Company’s booster…

On Dec. 13, 2021, Moderna announced an agreement with the Australian Government to build a state-of-the-art messenger RNA…

On Dec. 10, 2021, Moderna announced an amendment to its existing contract with Gavi, the Vaccine Alliance, to…

On Dec. 2, 2021, National Resilience and Harvard University announed they had established a five-year research and development…

On Dec. 1, 2021, Moderna announced a revised supply agreement with the UK government for up to 60…

On Nov. 19, 2021, the U.S. Food and Drug Administration (FDA) announced that it had extended the emergency…