ViiV Healthcare announced FDA approval for Rukobia (fostemsavir), a first-in-class treatment for HIV in adults
On Jul. 2, 2020, ViiV Healthcare, a GSK company, with Pfizer and Shionogi Limited, announced the FDA had…

On Jul. 2, 2020, ViiV Healthcare, a GSK company, with Pfizer and Shionogi Limited, announced the FDA had…
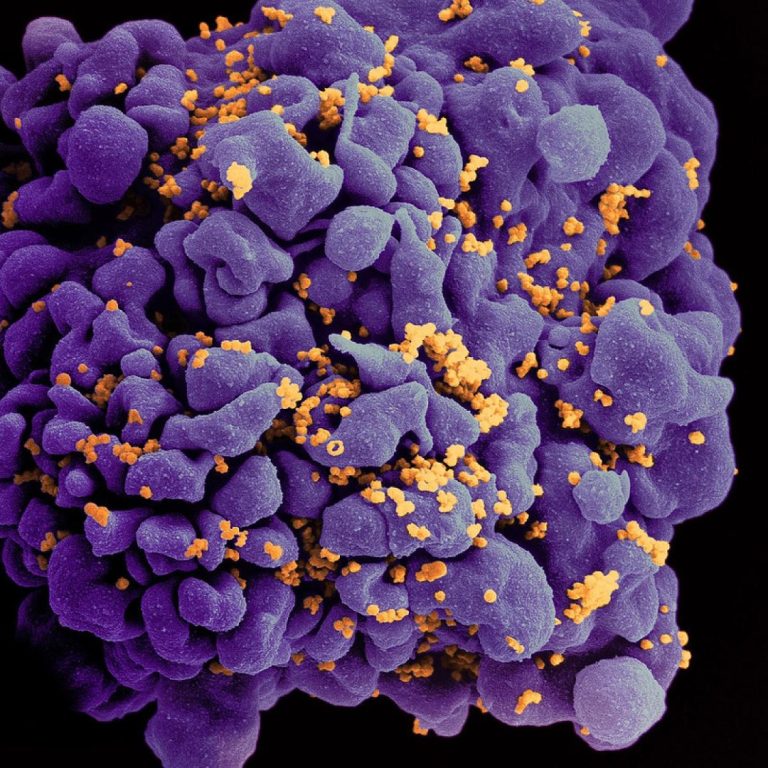
On Jun. 12, 2020, the U.S. Food and Drug Administration (FDA) approved ViiV Healthcare’s Tivicay (dolutegravir) tablets and…

On Jun. 12, 2020, ViiV Healthcare, a GSK company, with Pfizer and Shionogi Limited, announced the FDA had…
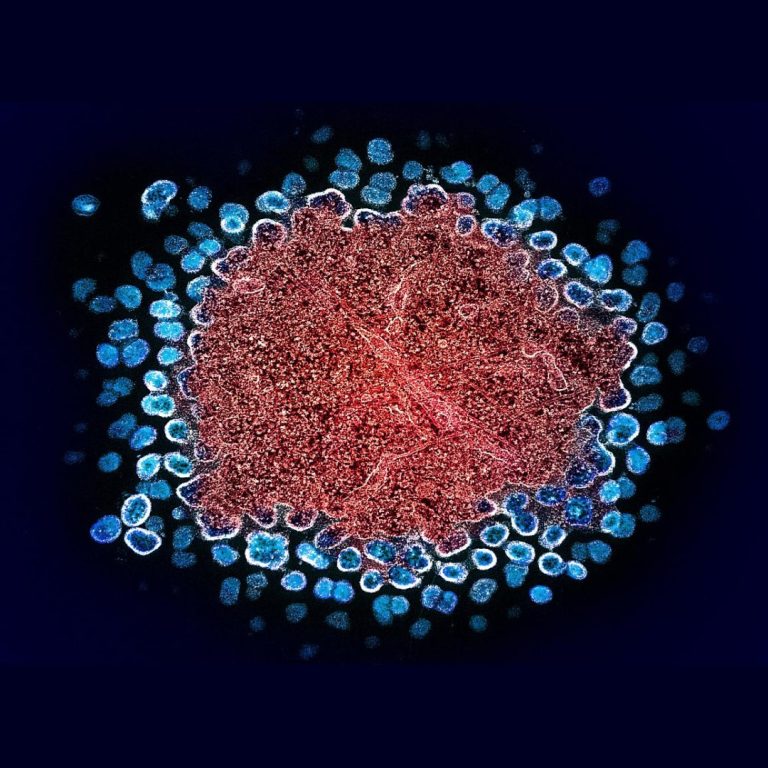
On Jun. 11, 2020, researchers have found that astrocytes, a type of brain cell can harbor HIV and…

On Jun. 8, 2020, Evotec announced that its Seattle-based subsidiary Just – Evotec Biologics had entered into an…

On May 26, 2020, the NIH announced a new study that provides an alternative treatment regimen that is…
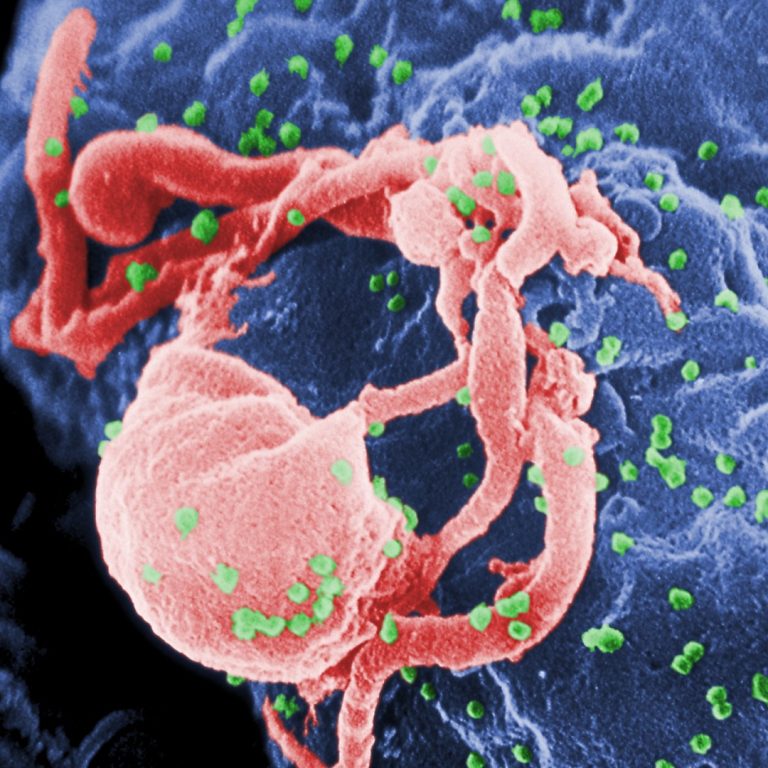
On May 18, 2020, an investigational long-acting form of the HIV drug cabotegravir injected once every 8 weeks…
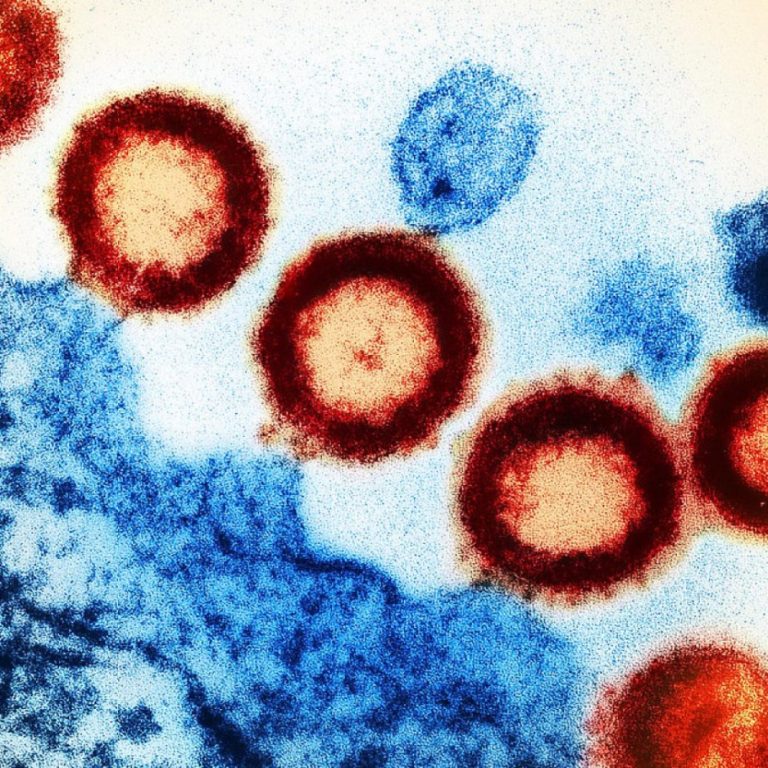
On Apr. 29, 2020, Scripps researchers announced they had developed a method for examining the machinery of HIV…

On Apr. 27, 2020, a novel means to prevent HIV infection was developed at the University of Nebraska…

On Mar. 25, 2020, a consortium of life sciences companies announced an important collaboration to accelerate the development,…

On Mar. 23, 2020, researchers from the University of Oxford announced they have launched a new clinical trial…
On Mar. 18, 2020, Immunology researchers at the University of Miami Miller School of Medicine announced a collaboration…

On Mar. 16, 2020, the Hormel Institute, University of Minnesota announced published research concerning remnants of ancient viruses…

On Mar. 11, 2020, the antiretroviral drugs dolutegravir and emtricitabine/tenofovir alafenamide fumarate (DTG+FTC/TAF) may comprise the safest and…
On Mar. 9, 2020, researchers from the National Institute of Allergy and Infectious Diseases (NIAID), part of the…

On Mar. 9, 2020, among African adolescent girls and young women who took HIV pre-exposure prophylaxis (PrEP) daily,…

On Feb. 4, 2020, the Fred Hutchinson Cancer Research Center announced that a promising HIV vaccine trial was…

On Jan. 8, 2020, Xencor announced a technology license agreement in which Gilead Sciences will access Xencor’s Xtend…

On Dec. 4, 2019, PATH announced that its Board of Directors had unanimously chosen global development leader Nikolaj…

On Nov. 6, 2019, Abbott announced that a team of its scientists identified a new subtype of the…

On Nov. 4, 2019, Chembio Diagnostics announced its SURE CHECK HIV Self-Test has received World Health Organization (WHO)…
On Nov. 4, 2016, the U.S. Centers for Disease Control and Prevention’s (CDC) Immunization Practices Advisory Committee (ACIP)…

On Oct, 26, 2016, a team of researchers at the University of Arizona and University of Cambridge in…

On Apr. 4, 2016, UNMC researchers, Tony Wilson, Ph.D., and Susan Swindells, M.B.B.S., working with a $2.2 million,…

On May 31, 2015, the New England Primate Research Center (NEPRC) closed its doors. During its five decades…

On Sept. 3, 2014, Oregon Health & Science University (OHSU) announced it had been awarded a $25 million…

On Oct. 22, 2013 Myron Cohen received the North Carolina Award for Science. Dr. Cohen is a University…

On Aug. 12, 2011, the National Institute on Drug Abuse (NIDA), part of NIH, announced that Dr. David…

On Jun. 11, 2011, the U.S. Centers for Disease Control and Prevention (CDC) marked the 30th year since…

On Dec. 20, 2011, the U.S. Food and Drug Administration (FDA) announced approval of the first and only…