Quest Diagnostics dedicated special hours for non-COVID-19 testing for older and Highly vulnerable patients
On Mar. 21, 2020, through its “Peace of Mind” program, Quest Diagnostics took extra measures to ensure individuals…

On Mar. 21, 2020, through its “Peace of Mind” program, Quest Diagnostics took extra measures to ensure individuals…

On Mar. 20, 2020, BioReference Labs, an OPKO Health company, announced a collaboration with the State of New…
On Mar. 20, 2020, Cardax released a white paper outlining the potential role of astaxanthin in the treatment…

On Mar. 20, 2020, PRA Health Sciences announced the commercial availability of the COVID-19 Monitoring Program, a mobile…
On Mar. 20, 2020, Bellerophon Therapeutics announced the U.S. Food and Drug Administration (FDA) had granted emergency expanded…

On Mar. 20, 2020, Chembio Diagnostics announced receipt of a $4 million order from Bio-Manguinhos for the purchase…

On Mar. 19, 2020, Todos Medical announced it had entered into an exclusive distribution agreement for the U.S….

On Apr. 1, 2020, Cumberland Pharma announced a national initiative to support hospitals and clinics that use Caldolor…

On Mar. 19, 2020, Mesa Biotech announced it has significantly expanded its capacity to test clinical samples for…

On Mar. 19, 2020, LabCorp announced it is dedicating the first hour of service each day at its…
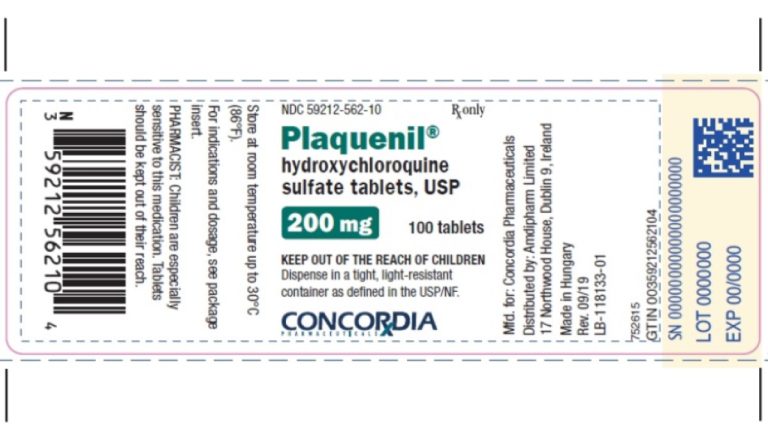
On Mar. 19, 2020, Teva announced the immediate donation of more than 6 million doses of hydroxychloroquine sulfate…

On Mar. 19, 2020, iBio announced its progress towards developing vaccine candidates for preventing infection from the SARS-CoV-2…

On Mar. 19, 2020, Clinical pathologists, infectious disease physicians and scientists at the UC Davis Medical Center, California…

On Mar. 19, 2020, Abbott announced the U.S. Food and Drug Administration (FDA) has issued Emergency Use Authorization (EUA)…
On Mar. 19, 2020, Roche announced it was working with the U.S. Food & Drug Administration (FDA) to…
On Mar. 18, 2020, Immunology researchers at the University of Miami Miller School of Medicine announced a collaboration…
On Mar. 18, 2020, Entos Pharmaceuticals, a health-care biotechnology company that develops new therapeutic compounds using the company’s…

On Mar. 18, 2020, scientists from the University of Oxford’s Engineering Science Department and the Oxford Suzhou Centre…

On Mar. 18, 2020, Abbott announced that the the U.S. Food and Drug Administration had issued Emergency Use…

On Mar. 18, 2020, Mateon Therapeutics reported significant progress in deploying its phase 3 clinical asset, OT-101, against…

On Mar. 18, 2020, Eli Lilly announced its scientists were partnering with the Indiana State Department of Health…

On Mar. 18, 2020, Emergent BioSolutions announced an agreement with Vaxart, whereby Emergent has agreed to utilize its…
On Mar. 18, 2020, Nike CEO John Donahoe and his wife, Eileen, along with Nike Chairman Mark Parker…

On Mar. 17, 2020, Quidel Corp. announced it had received Emergency Use Authorization (EUA) from the U.S. Food…

On Mar. 17, 2020, MannKind announced it was adjusting research and development resources that were reserved for its…
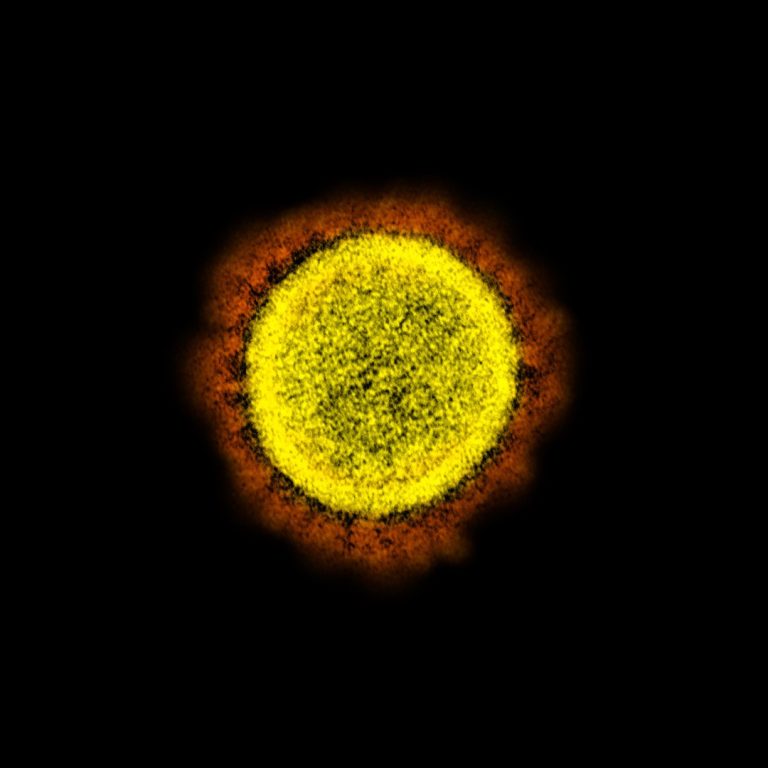
On Mar. 17, 2020, scientists reported the novel SARS-CoV-2 coronavirus that emerged in the city of Wuhan, China,…

On Mar. 17, 2020, Innovation Pharma announced details on the Material Transfer Agreement signed with a leading public…

On Mar. 17, 2020, BioReference Laboratories, an OPKO Health company, announced a collaboration with the New York City…
On Mar. 17, 2020, Pfizer and BioNTech announced the companies have agreed to a letter of intent regarding…

On Mar. 17, 2020, Regeneron announced that its scientists had isolated hundreds of virus-neutralizing, fully human antibodies from…