Mesa Biotech to develop molecular point-of-care diagnostic test for novel Coronavirus (COVID-19)
On Apr. 4, 2020, Mesa Biotech announced the addition of the novel coronavirus (SARS-CoV-2) to its active influenza…
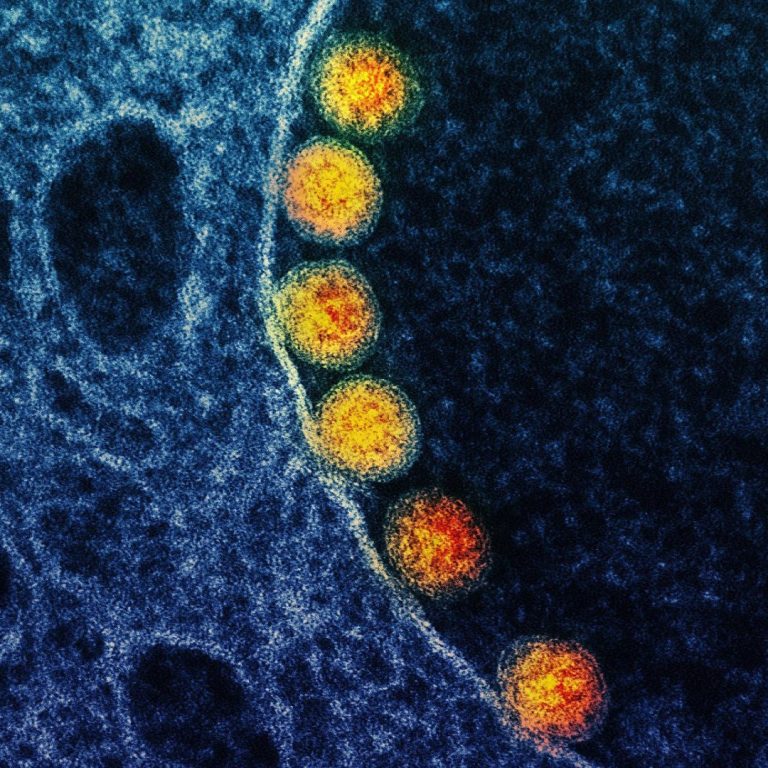
On Apr. 4, 2020, Mesa Biotech announced the addition of the novel coronavirus (SARS-CoV-2) to its active influenza…

On Apr. 3, 2020, the University of Minnesota announced it had created the stateメs fourth on-site testing facility…
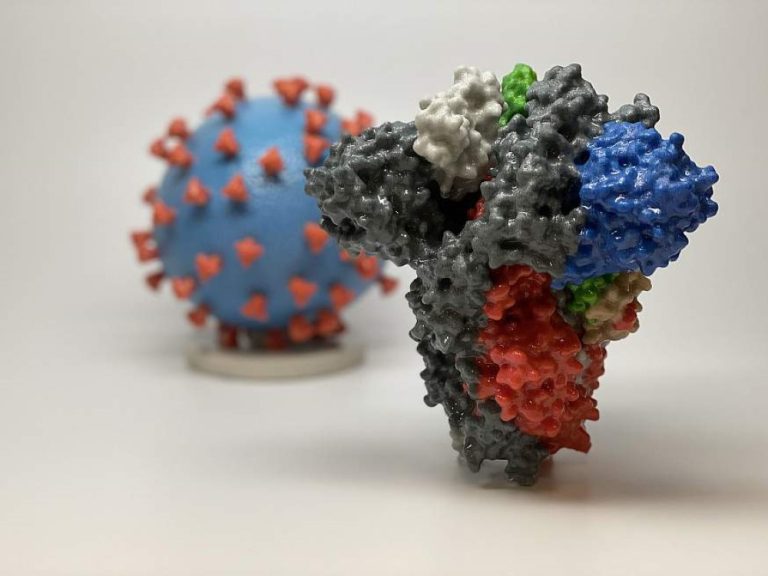
On Apr. 2, 2020, Vir Biotech and Alnylam Pharma announced an expansion to their broad multi-target existing collaboration…

On Apr. 2, 2020, Cerus announced that the International Society of Blood Transfusion (ISBT) Working Party on Global…

On Apr. 2, 2020, Vanda Pharmaceuticals announced initiation of clinical study, ODYSSEY VLY-686-3501, in hospitalized patients with COVID-19….

On Apr. 2, 2020, a team of scientists at Fred Hutchinson Cancer Research Center in Seattle began searching…

On Apr. 2, 2020, the FDAラin collaboration with the CDC, the Biodefense and Emerging Infections Research Resources Repository…

On Apr. 2, 2020, Novartis announced plans to initiate a phase III clinical trial in collaboration with Incyte…

On Apr. 2, 2020, an international team led by University of British Columbia (UBC) researcher Dr. Josef Penninger…
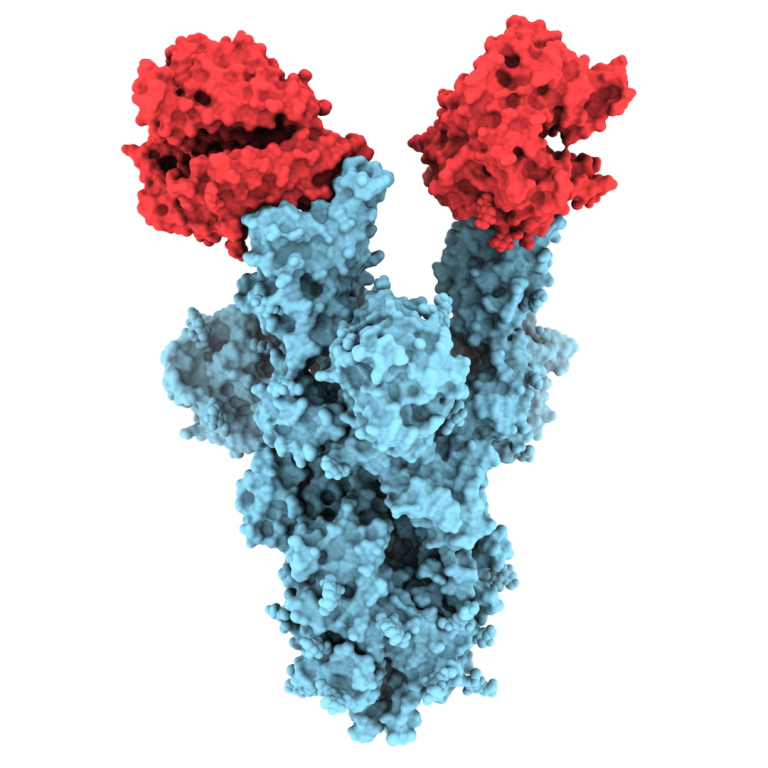
On Apr. 1, 2020, ImmunityBio, a privately held immunotherapy company within the NantWorks ecosystem of companies, announced it…

On Apr. 1, 2020, Innovation Pharma it had received data supporting Brilacidinメs direct inhibition of SARS-CoV-2, the novel…

On Apr. 1, 2020, a University of Washington Medicine led study of 24 of the most critically ill…

On Mar. 31, 2020, FUJIFILM announced initiation of a phase III clinical trial to evaluate the safety and…

On Mar. 31, 2020, Philips announced that it was fully committed to fulfill the contract with the U.S….

On Mar. 31, 2020, Cue Health announced that is had been awarded up to $30 million in base…
On Mar. 31, 2020, Mayo Clinic in Florida announced it had significantly ramped up efforts to process and…

On Mar. 31, 2020, Luminex announced it had received $642,450 in funding from the Biomedical Advanced Research and…

On Mar. 31, 2020, VBI Vaccines announced a collaboration with the National Research Council of Canada (NRC) to…

On Mar. 30, 2020, Humanigen announced that the company had submitted an initial protocol synopsis to the FDA…

On Mar. 30, 2020, researchers from the University of Minnesota broke new ground in understanding how SARS-CoV-2, the…

On Mar. 30, 2020, the Institute for Molecular Medicine in Finland (FIMM) announced it had launched a COVID-19…

On Mar. 30, 2020, Howard Hughes Medical Institute Investigator David Baker created new proteins tailor-made for specific tasks….

On Mar. 30, 2020, the Translational Genomics Research Institute (TGen) announced that it had joined the world-wide effort…

On Mar. 29, 2020, a study by UNMC/Nebraska Medicine/NSRI researchers provides new evidence of SARS-CoV-2 environmental contamination in…

On Mar. 27, 2020, University of Oxford researchers working in an unprecedented vaccine development effort to prevent COVID-19…
On Mar. 27, 2020, Luminex announced the U.S. Food and Drug Administration (FDA) issued an Emergency Use Authorization…

On Mar. 26, 2020, Thermo Fisher Scientific announced it had received the CE mark in the European Union…
On Mar. 26, 2020, physician-scientists at four University of California Health medical centers — UC San Diego Health,…
On Mar. 26, 2020, iBio announced that immunization studies for its SARS-CoV-2 Virus-Like Particle program (‘IBIO-200’) were proceeding…

On Mar. 25, 2020, Quidel received CE Mark for the Lyra SARS-CoV-2 assay, authorizing Quidel to market and…