Leiden Report Show Universities in China Dominate Top Ten Rankings
On Oct. 29, 2025, the CWTS Leiden ranking show that Harvard University dropped to third place in the…

On Oct. 29, 2025, the CWTS Leiden ranking show that Harvard University dropped to third place in the…
On Oct. 28, 2025, The Louisiana Illuminator reported that when there’s an outbreak of a vaccine-preventable disease, state…
As of Oct. 24, 2025, the South Carolina Department of Public Health (DPH) reported 33 cases of measles…
On Oct. 22, 2025, the South Carolina Department of Public Health (SCDPH) announced it is actively responding to…

On Oct. 21, 2025, the Oregon Department of Agriculture has warned that as wild birds begin to migrate…

On Oct. 20, 2025, Merck announced the start of construction for a $3 billion, 400,000-square-foot pharmaceutical manufacturing facility…

On Oct. 15, 2025, mRNA COVID-19 vaccination in the first trimester of pregnancy isn’t tied to an elevated…

On Oct. 14, 2025, Japan’s health authorities have declared an influenza epidemic, with thousands of people infected with…

On Oct. 7, 2025, the World Health Organization (WHO) launched the Global Clinical Trials Forum (GCTF), a global,…

On Oct. 2, 2025, the South Carolina Department of Public Health (DPH) has confirms a measles outbreak in…
On Oct. 1, 2025, Kaiser Permanente Washington announced results of the first study within a U.S.-based health system…

On Sept. 30, 2025, Zoetis announced that Dectomax®-CA1 Injectable is the first and only parasite control product to…
On Sept. 24, 2025, Indian drugmakers Dr Reddy’s Laboratories and Hetero Labs announced they will sell generic versions…

On Sept. 22, 2025, The Gates Foundation will give $912 million to the Global Fund to Fight AIDS,…

On Sept. 19, 2025, the state health officials of the members of the West Coast Health Alliance, including…

On Sept. 19, 2025, Health Secretary Robert F. Kennedy Jr.’s new vaccine advisers added confusion to this fall’s…
On Sept. 14, 2025, the World Health Organization announced that vaccination of frontline health workers and contacts of…
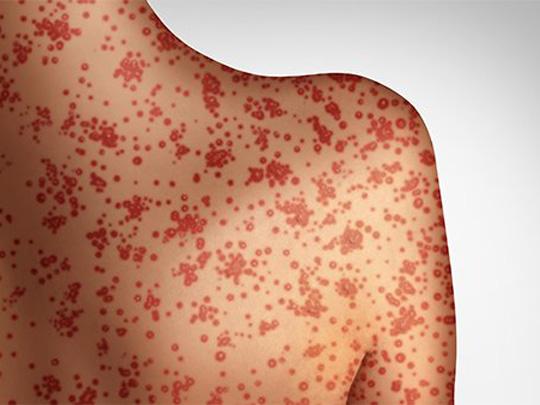
On Sept. 10, 2025, the Utah Department of Health and Human Services (UDOH) reported a potential large exposure…

On Sept. 9, 2025, Emergent BioSolutions announced that a contract modification has been executed in the amount of…

On Sept. 8, 2025, Pfizer and BioNTech announced positive topline results from an ongoing Phase 3 clinical trial…
On Sept. 5, 2025, the World Health Organization announced that it no longer considers the mpox outbreak in…

On Sept. 5, 2025, The Washington State of Health (DOH) issued a Standing Order for the COVID-19 vaccine,…

On Sept. 4, 2025, roughly 24 hours after the launch of the West Coast Health Alliance, Hawaii is…

On Sept. 4, 2025, Novavax announced a milestone payment from Takeda has been triggered by regulatory approval for Novavax’s…
On Sept. 3, 2025, California Governor Gavin Newsom, Oregon Governor Tina Kotek, and Washington Governor Bob Ferguson announced…

On Sept. 3, 2025, Avian flu has hit turkey farms in North Dakota and South Dakota, the first…
On Sept. 2, 2025, Emergent BioSolutions announced it had received a $30 million contract modification from the Biomedical Advanced…
On Aug. 28, 2025, scientists at MIT’s Computer Science and Artificial Intelligence Laboratory (CSAIL) and the MIT Abdul…

On Aug. 27, 2025, the U.S. Centers for Disease Control and Prevention (CDC) Director Susan Monarez has left…

On Aug. 27, 2025, Novavax announced that the U.S. Food and Drug Administration (FDA) has approved the Nuvaxovid™ 2025-2026…