The FDA permitted marketing of first game-based digital therapeutic to improve attention function in children with ADHD
On Jun. 15, 2020, the U.S. Food and Drug Administration (FDA) permitted marketing of the first game-based digital…

On Jun. 15, 2020, the U.S. Food and Drug Administration (FDA) permitted marketing of the first game-based digital…

On Jun. 15, 2020, Humanigen announced data on the first clinical use of lenzilumab in 12 COVID-19 patients….

On Jun. 15, 2020, SIGA Technologies announced the U.S. Department of Defense (DoD) increased research and development funding…
On Jun. 15, 2020, the U.S. Food and Drug Administration (FDA) warned health care providers about a newly…

On Jun. 15, 2020, the U.S. Food and Drug Administration (FDA) revoked the emergency use authorization (EUA) that…
On Jun. 13, 2020, Dr. Reddy’s Laboratories announced that it had entered into a non-exclusive Licensing Agreement with…

On Jun. 12, 2020, the California Institute for Regenerative Medicine (CIRM) awarded $349,999 to Dr. Vaithilingaraja Arumugaswami at…
On Jun. 12, 2020, the U.S. Food and Drug Administration (FDA) approved ViiV Healthcare’s Tivicay (dolutegravir) tablets and…

On Jun. 11, 2020, Bristol-Myers Squibb announced that Opdivo (nivolumab) was approved by the FDA for the treatment…
On Jun. 11, 2020, researchers have found that astrocytes, a type of brain cell can harbor HIV and…

On Jun. 11, 2020, BioCryst Pharmaceuticals announced new data published in Science Translational Medicine that show, in a…
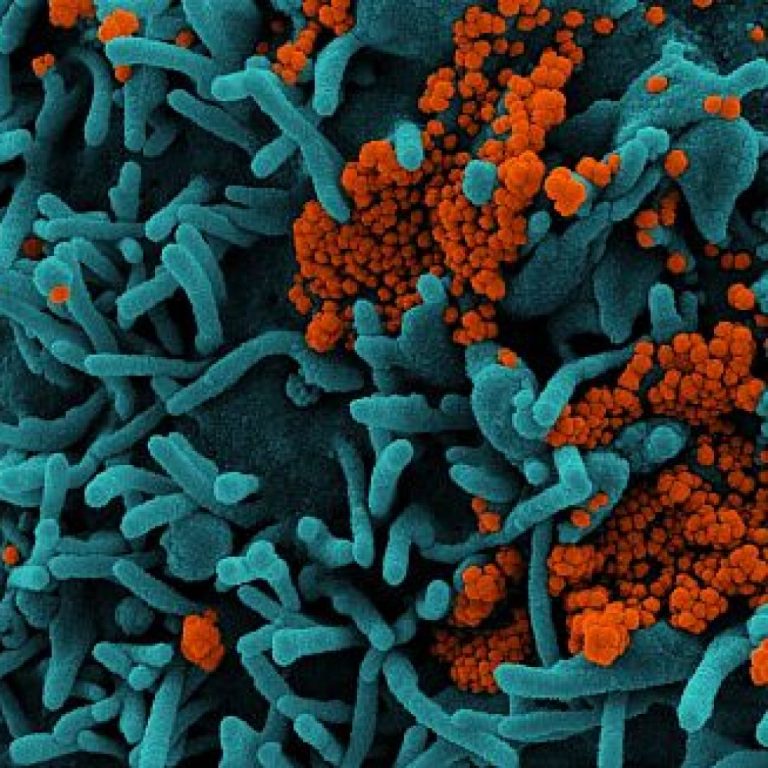
On Jun. 11, 2020, Regeneron announced that Science had accepted for publication two papers describing the creation of…

On Jun. 10, 2020, researchers funded by the National Institutes of Health (NIH) launched an effort to evaluate…

On Jun. 10, 2020, Washington University School of Medicine in St. Louis reported they had developed a mouse…

On Jun. 10, 2020, OraSure Technologies announced it had been awarded a $629,217 contract from the Biomedical Advanced…

On Jun. 10, 2020, RedHill Biopharma announced it had submitted a Clinical Trial Application (CTA) with the Ministry…
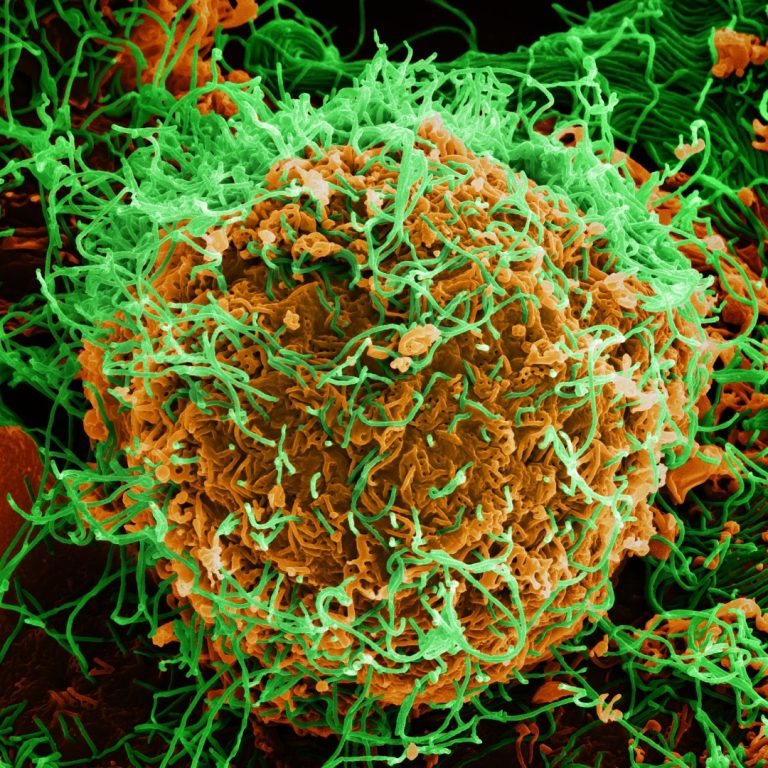
On Jun. 9, 2020, new genetic sequence analysis by the Democratic Republic of the Congo’s (DRC) National Institute…

On Jun. 9, 2020, an international gnomAD team of over 100 scientists released its first set of discoveries…

On Jun. 8, 2020, Aimmune Therapeutics announced two-year data that suggested long-term efficacy of daily treatment with PALFORZIA…

On Jun. 8, 2020, researchers at the National Human Genome Research Institute announced they had discovered clues to…

On Jun. 5, 2020, the Alberta Precision Laboratories (APL) COVID-19 Biorepository was announced to ensure researchers have the…
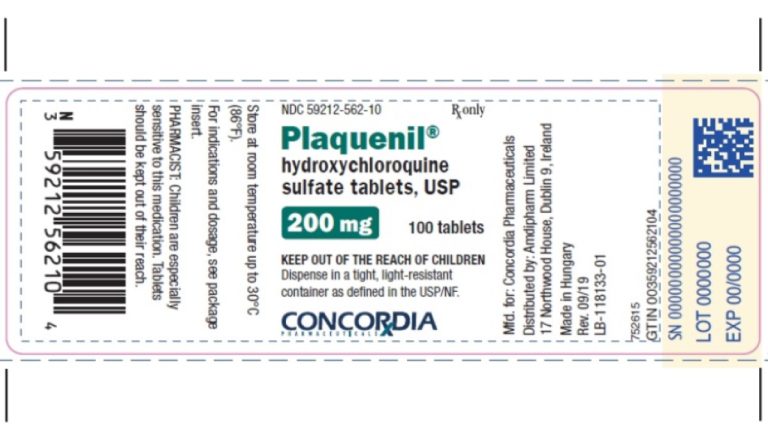
On Jun. 5, 2020, the Chief Investigators of the randomised evaluation of COVid-19 thERapY (RECOVERY) trial on hydroxychloroquine…
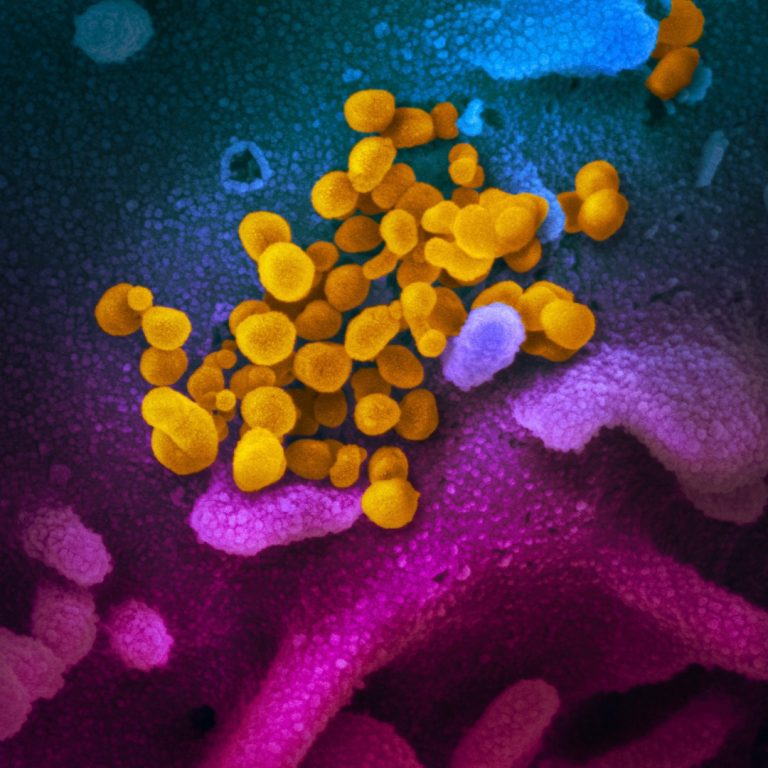
On Jun. 5, 2020, Sorrento announced it had completed a preclinical batch of the STI-4398 (COVIDTRAP) protein and…

On Jun. 5, 2020, BioSig Technologies and its subsidiary, ViralClear Pharma, announced it had expanded its patient enrollment…

On Jun. 4, 2020, AstraZeneca announced the next steps in its commitment to broad and equitable global access…
On Jun. 1, 2020, Gilead Sciences announced topline results from the Phase 3 SIMPLE trial in hospitalized patients…

On Jun. 1, 2020, Emergent BioSolutions announced it had been issued a task order under an existing contract…
On Jun. 1, 2020, Anixa Biosciences and OntoChem announced they had synthesized four potential Covid-19 compounds that will…

On Jun. 1, 2020, Tonix Pharmaceuticals announced an agreement whereby FUJIFILM Diosynth Biotechnologies will provide contract manufacturing and…

On Jun. 1, 2020, SIGA Technologies announced its first international delivery of TPOXX (tecovirimat), with 2,500 courses delivered…