Moderna announced mRNA-1345, an investigational RSV Vaccine, met primary efficacy endpoints in phase 3 trial
On Jan. 17, 2023, Moderna announced positive topline data from its ConquerRSV Phase 3 pivotal efficacy trial of…

On Jan. 17, 2023, Moderna announced positive topline data from its ConquerRSV Phase 3 pivotal efficacy trial of…

On Jan. 17, 2023, the Montana Fish and Wildlife Commission (FWP) reported that juvenile grizzly bears tested positive…

On Jan. 11, 2023, Roche announced treatment of uncomplicated influenza and for post-exposure prophylaxis of influenza. Post-exposure prophylaxis…
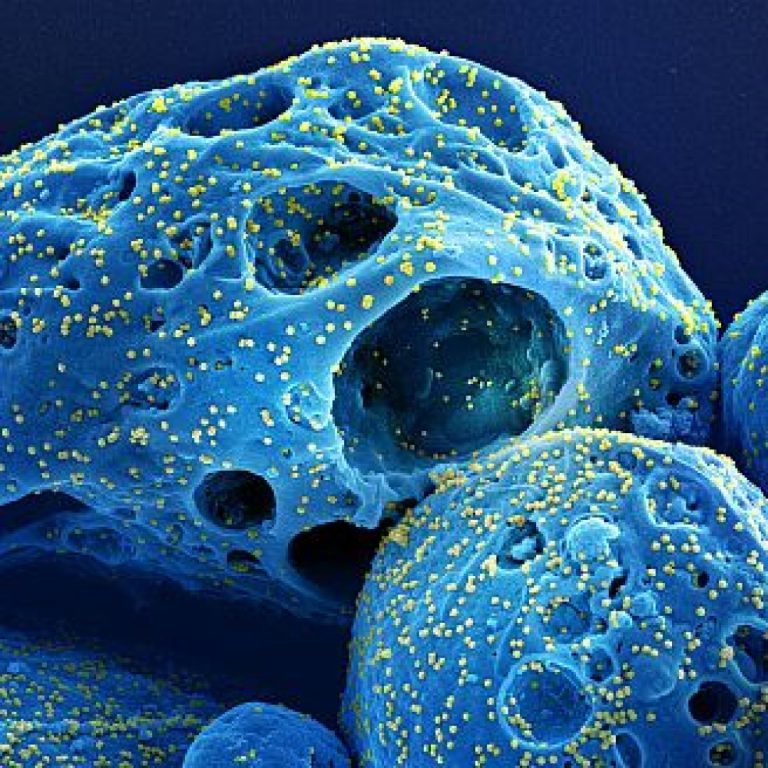
On Jan. 10, 2023, the U.S. National Institutes of Health (NIH) reported that antiviral treatments can help reduce…

On Jan. 5, 2023, Novavax announced that Spain’s Public Health Commission, Italy’s Ministry of Health, and France’s Haute…

On Jan. 1, 2023, a study published in the Lancet reported that concentrations of antibiotic residues found in…

On Dec. 21, 2022, Moderna announced the the finalization of a strategic partnership with the United Kingdom (UK)…

On Nov. 12, 2022, the World Health Organization (WHO) issued a Disease Outbreak News (DON) on the circulating…

On Dec. 16, 2022, Moderna announced that the European Medicines Agency’s Committee for Medicinal Products for Human Use…
On Dec. 16, 2022, Novavax announced that the Standing Committee on Vaccination (STIKO) in Germany had expanded its…
On Dec. 15, 2022, the University of Oxford’s Ebola vaccine candidate has been shipped to Uganda, just 80…

On Dec. 12, 2022, the Oregon Department of Agriculture and the U.S. Department of Agriculture’s Animal Plant Health…

On Dec. 13, 2022, Texas Biomed announced that it had been designated as a prime contractor by the…

On Dec. 11, 2022, Novavax announced that the Switzerland’s Federal Office of Public Health and Francees Haute Autorite…

On Dec. 9, 2022, the World Health Organization (WHO) announced that the first doses of one of the…

On Dec. 8, 2022, Moderna announced it had received emergency use authorization from the U.S. Food and Drug…
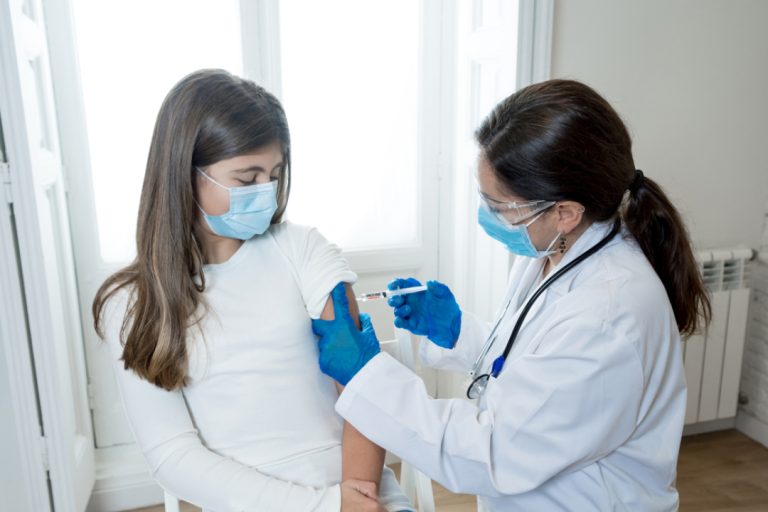
On Dec. 5, 2022, the Fred Hutchinson Cancer Center announced that according to a new meta-analysis women were…

On Dec. 5, 2022, Pfizer and BioNTech announced that the companies had submitted an application to the U.S….

On Dec. 2, 2022, the Oregon Department of Agriculture (ODA) and the U.S. Department of Agriculture’s (USDA) Animal…

On Nov. 29, 2022, Novavax announced that the World Health Organization had issued an updated Emergency Use Listing…

On Nov. 27, 2022, it was reported a parasite that infects up to one-third of people around the…

On Nov. 18, 2022, the World Health Organisation for Animal Health (WHO) announced that Peru had reported its…

On Nov. 18, 2022, Novavax announced that Health Canada had granted expanded authorization for Nuvaxovid (COVID-19 Vaccine (Recombinant…

On Nov. 15, 2022, Moderna reported findings from a Phase II/III clinical trial where bivalent Omicron-targeting booster candidates,…
On Nov. 9, 2022, Novavax announced that the Medicines and Healthcare products Regulatory Agency (MHRA) in the United…
On Nov. 8, 2022, Novavax announced topline results from its Phase 3 Boosting Trial for the SARS-CoV-2 rS…

On Nov. 5, 2022, the U.S. Department of Agriculture’s (USDA) Animal Plant Health Inspection Service (APHIS) confirmed the…

On Nov. 3, 2022, Health Canada authorized an adapted version of the Moderna Spikevax COVID-19 vaccine that targets…

On Nov. 2, 2022, Hologic announced that it had been awarded a $19 million contract from the Biomedical…

On Oct. 31, 2022, Moderna announced that it had received approval from the Ministry of Health, Labour and…