U.S. FDA permitted marketing of first COVID-19 at-home test using traditional premarket review process
On Jun. 6, 2023, the U.S. Food and Drug Administration (FDA) granted granted marketing authorization for the Cue COVID-19…
On Jun. 6, 2023, the U.S. Food and Drug Administration (FDA) granted granted marketing authorization for the Cue COVID-19…

On May 31, 2023, Pfizer announced that the U.S. Food and Drug Administration (FDA) had approved ABRYSVO (Respiratory…

On May 30, 2023, the United Kingdom of Great Britain and Northern Ireland reported to the World Health…

On May 26, 2023, Novavax announced that Nuvaxovid (NVX-CoV2373) had been recommended for full Marketing Authorization (MA) for…

On May 25, 2023, the U.S. Food and Drug Administration (FDA) approved the oral antiviral Paxlovid (nirmatrelvir tablets…

On May 15, 2023, Brazil, the world’s top chicken exporter, for the first time confirmed Highly Pathogenic Avian…

On May 12, 2023, the U.S. Departments of Agriculture Animal (USDA) and Plant Health Inspection Service (APHIS) announced…
On May 11, 2023, the World Health Organization announced it was ending a 10-month-long global health emergency for…
On May 5, 2023, the National Institutes of Health reported that a study had identified features of Long…
On May 5, 2023, the World Health Organization (WHO) ended its declaration of COVID-19 as a global health…
On May 5, 2023, France reported nine cases of severe neonatal sepsis associated with hepatic impairment and multi-organ failure…

On May 3, 2023, the U.S. FDA approved GlaxoSmithKline’s Arexvy as the first respiratory syncytial virus (RSV) vaccine…
On May 2, 2023, the U.S. Preventive Services Task Force (USPSTF) published a final recommendation statement on screening…

On May 2, 2023, the U.S. Food and Drug Administration (FDA) announced that it was taking additional steps…
On Apr. 27, 2023, a National Institute of Allergy and Infectious Diseases (NIAID) -led Phase 2 trial compared two…
On Apr. 24, 2023, the U.S. Food and Drug Administration (FDA) authorized Status COVID-19 Antigen Rapid Test for…

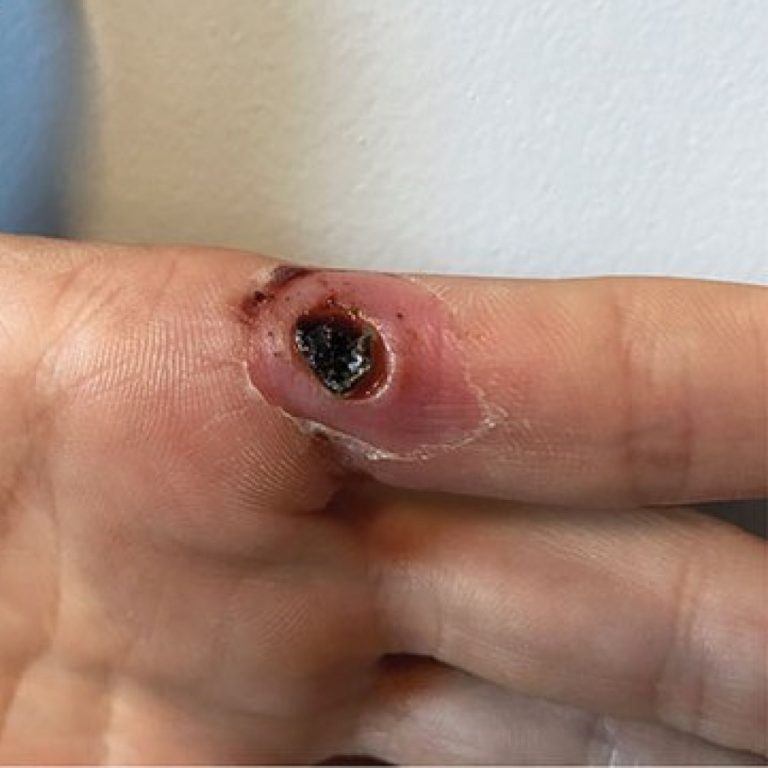
On Apr. 17, 2023, the World Health Organization (WHO) announced that six additional laboratory-confirmed cases of Marburg virus…
On Apr. 10, 2023, the U.S. Department of Health and Human Services (HHS) announced it was spending over…

On Apr. 7, 2023, the U.S. National Park Service reported Highly Pathogenic Avian Influenza (HPAI) had been confirmed…

On Apr. 5, 2023, Aberdeen Proving Ground South, formerly known as Edgewood, celebrated the opening of the Department…

On Apr. 5, 2023, Pfizer announced that experimental respiratory syncytial virus (RSV) vaccine was 82% effective in preventing…
On Apr. 4, 2023, National Institutes of Health (NIH) funded research was announced from the University of Rochester…

On Apr. 4, 2023, NanoViricides announced that it had executed a license agreement with Karveer Meditech, Kolhapur, India,…

On Mar. 31, 2023, the Oregon Department of Agriculture and the United States Department of Agriculture’s Animal and…

On Mar. 30, 2023, Moderna and the Government of the Republic of Kenya announced they had finalized an…

On Mar. 29, 2023, the mortality gap between the United States and other high-income nations substantially expanded during…

On Mar. 27, 2023, the National Health Commission of the People’s Republic of China reported a confirmed case…

On Mar. 24. 2023, antibiotics do not reduce the risk of dying in adults hospitalized with COVID-19, the…
On Mar. 23, 2023, a study published in Scientific Reports warned that vibrio vulnificus is an increasingly recognized…

On Mar. 23. 2023, researchers at Washington University School of Medicine in St. Louis and the Veterans Affairs…