Cue’s Molecular, point-of-care COVID-19 tests available under HHS and DoD pilot program
On Jan. 19, 2021, Cue Health announced that its molecular, point-of-care COVID-19 Tests were being distributed to five…

On Jan. 19, 2021, Cue Health announced that its molecular, point-of-care COVID-19 Tests were being distributed to five…

On Jan. 15, 2021, scientists at Washington University School of Medicine in St. Louis announced that that a…

On Jan. 14, 2021, Mammoth Biosciences announced that it had secured a subcontract with MRIGlobal, the prime contractor…

On Jan. 12, 2021, the Tufts Center for the Study of Drug Development (Tufts CSDD) reported that ever…
On Jan. 11, 2021, AXIM Biotechnologies announced that it had released a preprint of its manuscript describing the…

On Jan. 8, 2021, the American Cancer Society reported that the death rate from cancer in the U.S….

On Jan. 8, 2021, Pfizer and BioNTech announced results from an in vitro study conducted by Pfizer and…

On Jan. 7, 2021, Twist Bioscience announced it would supply the U.S. Centers for Disease Control and Prevention…
On Jan. 5, 2021, Illumina and Helix announced a collaboration to augment national surveillance infrastructure in the U.S….
On Dec. 21, 2020, the WHO reported that authorities of the United Kingdom of Great Britain and Northern…

On Dec. 21, 2020, Aimmune Therapeutics announced that the European Commission (EC) had approved PALFORZIA [defatted powder of…

On Dec. 21, 2020, Quidel announced that it had received Emergency Use Authorization from the U.S. Food and…

On Dec. 18, 2020, the the U.S. Food and Drug Administration (FDA) approved AstraZeneca’s Tagrisso (osimertinib) as the…

On Dec. 16, 2020, BD (Becton, Dickinson) announced that it had received pandemic orders for needles and syringes…
On Dec. 15, 2020, AXIM Biotechnologies announced the development and patent filing for an enzyme-linked immunosorbent assay (ELISA)-based…

On Dev. 11, 2020, the University of Oxford reported that data from the National COVID-19 Infection Survey, done…

On Dec. 4, 2020, the U.S. Food and Drug Administration (FDA) authorized the first diagnostic test for at home collection…
On Dec. 3, 2020, AXIM Biotechnologies announced the development and patent filing for an enzyme-linked immunosorbent assay (ELISA)-based…

On Dec. 2, 2020, the University of Oregon opened the Phil and Penny Knight Campus for Accelerating Scientific…
On Dec. 2, 2020, BD (Becton, Dickinson) announced plans to invest approximately $1.2 billion over a 4-year period…

On Nov. 30, 2020, Moderna announced that the primary efficacy analysis of the Phase 3 study of mRNA-1273…

On Nov. 20, 2020, the ‘European Food Safety Authority reported that within the past month more than 300…
On Nov. 19, 2020, University of Oxford researchers announced that research into the HIV-1 virus had shed light…

On Nov. 19, 2020, Cue Health announced that, as of November 9, the U.S. Department of Health and…
On Nov. 19, 2020, XBiotech announced data for its breakthrough candidate therapy for treating infections of influenza and…

On Nov. 19, 2020, Dallas-based company, Worlds Inc., the U.S. Air Force and Texas A&M University announced a…

On Nov. 18, 2020, Oregon Health & Science University (OHSU) announced an initiative that will attempt to discern…
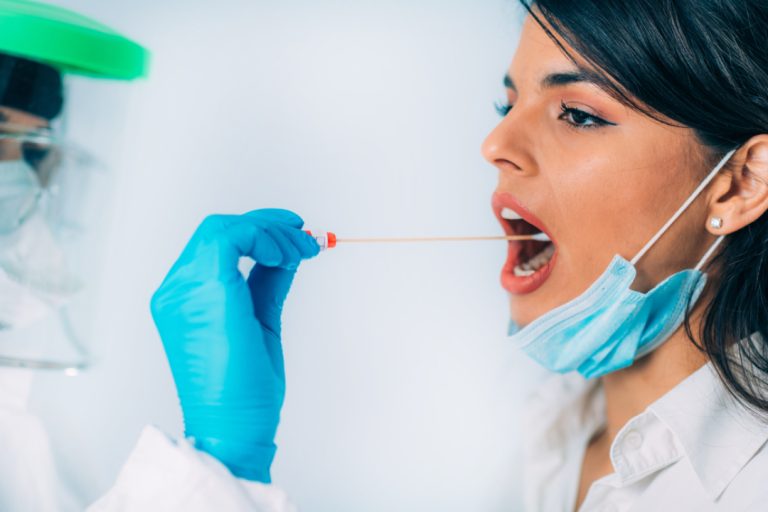
On Nov. 16, 2020, bioMerieux announced the expansion of its ARGENE range for the detection of SARS-CoV-2. As…

On Nov. 16, 2020, the UK Department for Health and Social Care Testing Innovation Fund announced £12.2M funding…

On Nov. 16, 2020, Tonix Pharmaceuticals announced preliminary results following vaccination of non-human primates with TNX-1800 (modified horsepox…