Moderna and the European Commission agreed on amendment to COVID-19 vaccine supply agreement
On Jun. 2, 2022, Moderna announced an agreement with the European Commission (EC) to amend their originally agreed…

On Jun. 2, 2022, Moderna announced an agreement with the European Commission (EC) to amend their originally agreed…
On Jun. 2, 2022, Novavax announced the submission of a request to the Medicines and Healthcare products Regulatory…

On Jun. 2, 2022, the U.S. Department of Agriculture’s Animal and Plant Health Inspection Service confirmed the presence…

On Jun. 2, 2022, the National Institutes of Health (NIH) reported that the Age-Related Eye Disease Studies (AREDS…

On May 31, 2022, Moderna and Takeda announced the transfer of the marketing authorization for Moderna’s COVID-19 vaccine…

On May 31, 2022, Novavax announced the initiation of its Phase 3 strain change trial to determine if…
On May 30, 2022, The Centre for Health Protection (CHP) of the Department of Health received notification from…

May 27, 2022, the National Cancer Institute (NCI) truncated the just-renewed five-year Cancer Center Support Grant held by…

On May 25, 2022, Novavax announced it was participating in a stage of the COVID-19 Vaccine Schedule Combinations…
On May 21, 2022, the World Health Organization (WHO) reported that since May 13, 2022, cases of mpox…

On May 20, 2022, Novavax announced the submission of a request to the European Medicines Agency (EMA) to…

On May 18, 2022, Moderna and the nonprofit scientific research organization IAVI announced that the first participant screenings…

On May 18, 2022, the U.S. Department of Agriculture’s (USDA) Animal and Plant Health Inspection Service (APHIS) confirmed…
On May 18, 2022, LabCorp announced the receipt of Emergency Use Authorization (EUA) from the U.S. Food and…
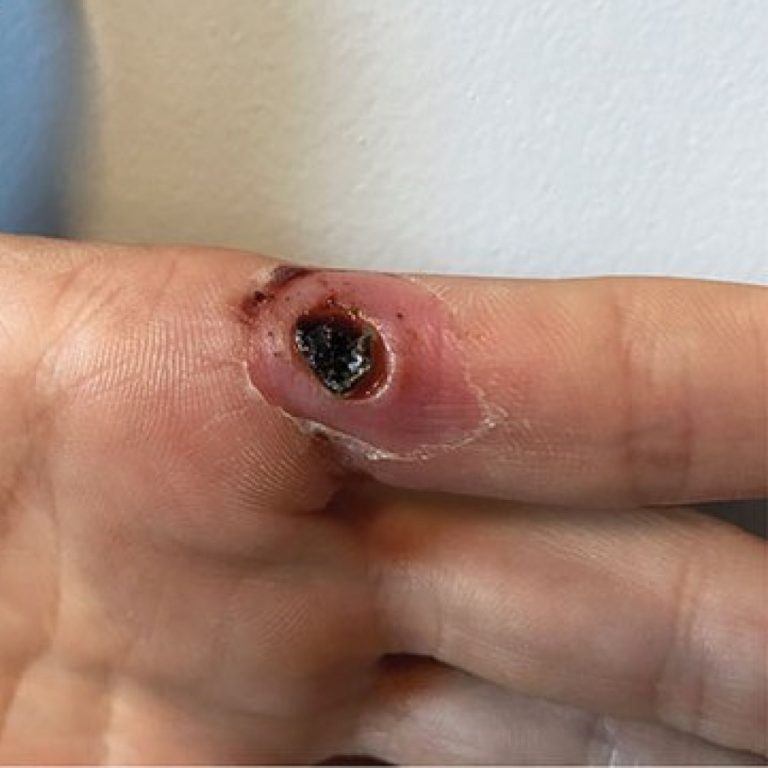
On May 18, 2022, scientists at the Centers for Disease Control and Prevention (CDC) announced that they were…

On May 13, 2022, the Tabula Sapiens Consortium announced it had published a molecular reference atlas for more…

On May 13, 2022, Novavax announced the submission of a request for emergency use authorization to Taiwan’s Food…
On May 11, 2022, Kaiser Permanente Washington Health Research Institute reported that new treatments for people at high…
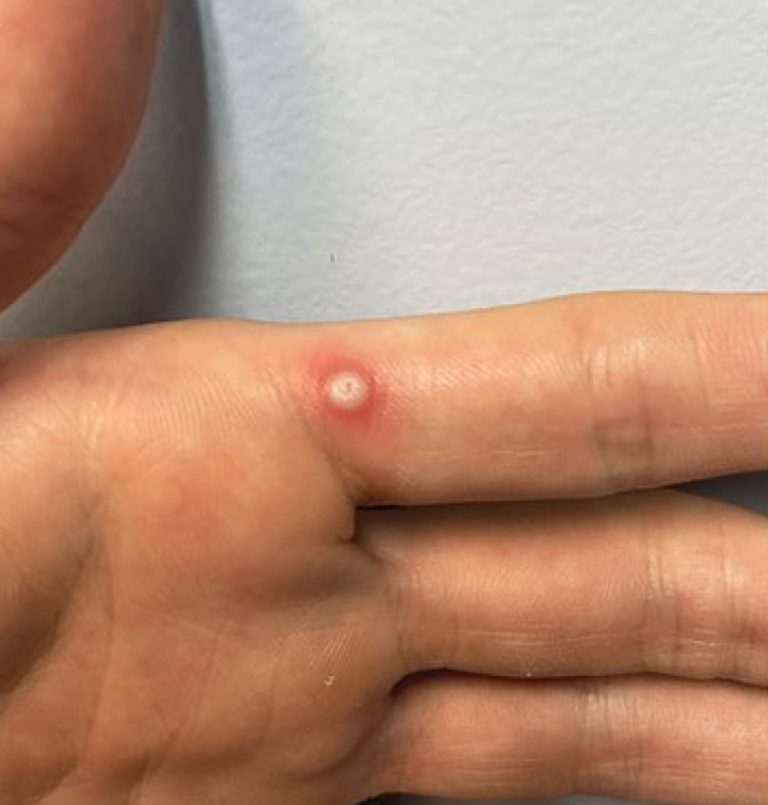
On May 7, 2022, the World Health Organization (WHO) was informed of a confirmed case of Mpox in…

On May 6, 2022, Novavax announced the submission of variations to the Australian Therapeutic Goods Agency (TGA) and…

On May 6, 2022, the Oregon Department of Agriculture (ODA) and United States Department of Agriculture (USDA) announced…
On May 6, 2022, Novavax announced that deliveries of it’s COVID-19 vaccine continued around the world and the…

On May 4, 2022, Oxitec announced approval from the Florida Department of Agriculture and Consumer Services, including reviews…
On May 2, 2022, Sorrento announced its Phase I study of intranasal STI-919 OVISHIELDTM) had been fully enrolled…

On Apr. 30, 2022, the U.S. Department of Agriculture’s (USDA) Animal and Plant Health Inspection Service (APHIS) confirmed…

On Apr. 29, 2022, Moderna announced its plan to build a state-of-the-art mRNA vaccine manufacturing facility in Quebec…

On Apr. 29, 2022, the U.S. Department of Agriculture’s (USDA) Animal and Plant Health Inspection Service (APHIS) confirmed…

On Apr. 28, 2022, Moderna announced that it had submitted a request for emergency use authorization (EUA) for…

On Apr. 28, 2022, the Ministry of Health of the Democratic Republic of the Congo declared an outbreak…

On Apr. 28, 2022, Centers for Disease Control and Prevention (CDC) reported that a person had tested positive…