Experts identifed steps to expand and improve antibody tests in COVID-19 response
On Jun. 26, 2020, more than 300 scientists and clinicians from the federal government, industry and academia published…

On Jun. 26, 2020, more than 300 scientists and clinicians from the federal government, industry and academia published…

On Jun. 26, 2020, the COVID-19 Therapeutics Accelerator donors and partners announced the formation of the International COVID-19…

On Jun. 26, 2020, Vaxart announced that its oral COVID-19 vaccine had been selected to participate in a…

On Jun. 26, 2020, the California Institute for Regenerative Medicine (CIRM) awarded $750,000 to Dr. Xiaokui Zhang at…

On Jun. 25, 2020, the University of Alberta (U of A) announced a study that will analyze thousands…

On Jun. 25, 2020, Entos Pharmaceuticals announced the selection of two lead candidates for a pan-coronavirus Fusogenix DNA…

On Jun. 25, 2020, the World Health Organization (WHO) announced the end of the 10th outbreak of Ebola…

On Jun. 25, 2020, the U.S. Food and Drug Administration (FDA) approved Fintepla (fenfluramine), a Schedule IV controlled…

On Jun. 25, 2020, the Government of Canada announced the results of that funding competition: an investment of…

On Jun. 25, 2020, researchers at the University of British Columbia announced they had received a combined total…

On Jun. 25, 2020, Vaxart announced it had signed a Memorandum of Understanding with Attwill Medical Solutions Sterilflow,…

On Jun. 25, 2020 Moderna announced a collaboration for large-scale, commercial fill-finish manufacturing of Moderna’s mRNA-based COVID-19 vaccine…

On Jun. 25, 2020, in support of Mayo Clinic’s digital health and practice transformation initiatives, the Mayo Clinic…

On Jun. 25, 2020, LabCorpᆴ announced the launch of a new test that can be used to assess…
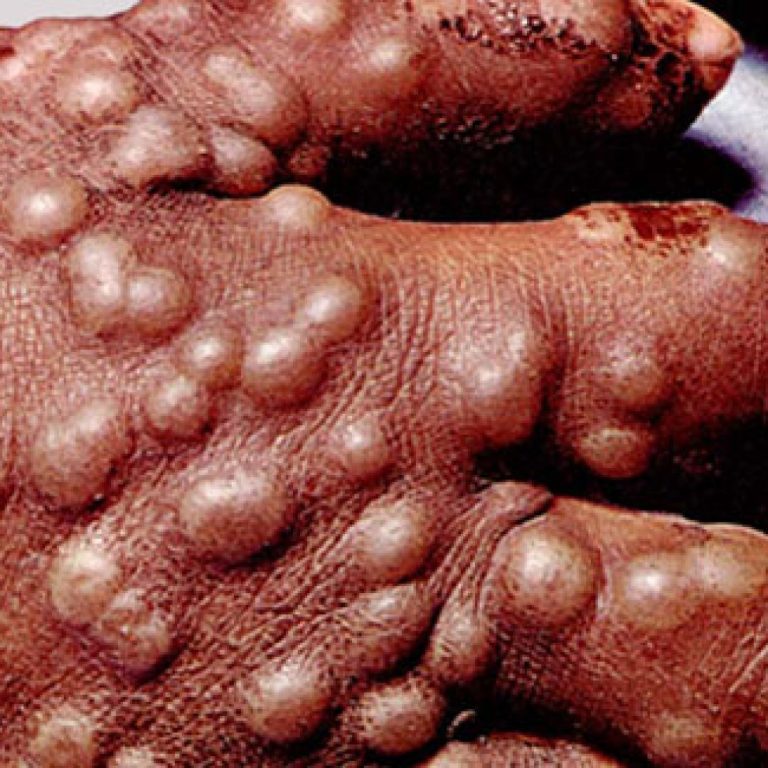
On Jun. 25, 2020, SIGA Technologies announced the deliveries of oral TPOXX (tecovirimat) to the U.S. Department of…
On Jun. 25, 2020, the Allen Institute announced a new a new $40.5 million collaborative research center in…
On Jun. 24, 2020, RedHill Biopharma announced that results from the treatment of the first severe COVID-19 patients…

On Jun. 24, 2020, HHMI investigators reported that data from COVID-19 cases around the world suggest that the…

On Jun. 24, 2020, Symbiosis Pharmaceutical Services, an India-based specialist provider of pharmaceutical and vaccine development services, announced…

On Jun. 24, 2020, the University of Oxford that the COVID-19 National DiagnOstic Research and Evaluation Platform (CONDOR)…

On Jun. 24, 2020, Sinovac Biotech announced the China National Medical Products Administration (NMPA) issued a product license…

On Jun. 24, 2020, Oncotelic, a wholly owned subsidiary of Mateon Therapeutics, announced that IBM has granted access…

On Jun. 24, 2020, iBio announced that IBM Watson Health has selected iBio to receive 18 months of…

On Jun. 24, 2020, a team of scientists at Texas A&M University announced it is working with iBio…

On Jun. 23, 2020, Mateon Therapeutics announced it had selected IQVIA to manage C001, a Phase 2 randomized,…

On Jun. 23, 2020, scientists at Scripps Research reported that some common strains of influenza have the potential…

On Jun. 23, 2020, Takeda Pharmaceutical announced that the FDA had approved the company’s submission for its biologics…

On Jun. 23, 2020, studies in the Netherlands and other places have shown that the presence of SARS-Coronavirus-2,…

On Jun. 23, 2020, Sanofi and Translate Bio announced they had agreed to expand their existing 2018 collaboration…

On Jun. 23, 2020, INOVIO announced it had received $71 million funding from the U.S. Department of Defense…