Aptima HIV-1 Quant Dx assay receivds additional FDA approval for use as aAid in diagnosis of HIV infection
On Nov. 30, 2020, Hologic announced that the FDA had approved a diagnostic claim for its HIV-1 (human…

On Nov. 30, 2020, Hologic announced that the FDA had approved a diagnostic claim for its HIV-1 (human…
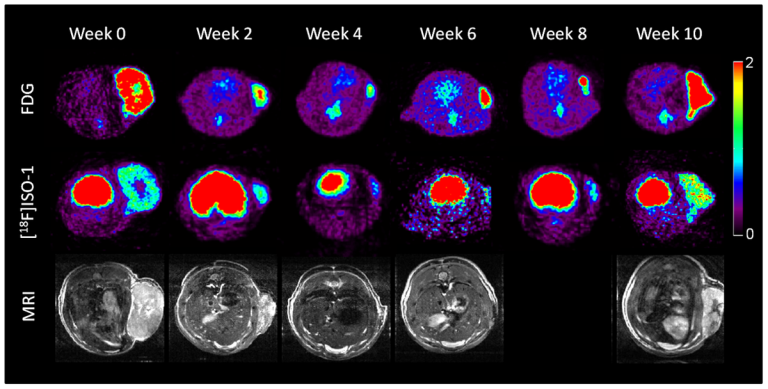
On Nov. 19, 2020, in a comprehensive analysis of patients with cancer who had exceptional responses to therapy,…

On Nov. 19, 2020, ApiJect Systems, a public benefit corporatio, announced that it had been approved by the…

On Nov. 19, 2020, F4 Pharma announced the inclusion of the first patient with severe COVID-19 in a…

On Nov. 19, 2020, the University of Oxford announced that the ChAdOx1 nCov-2019 coronavirus vaccine had been shown…
On Nov. 19, 2020, XBiotech announced data for its breakthrough candidate therapy for treating infections of influenza and…

On Nov. 19, 2020, Cue Health announced that, as of November 9, the U.S. Department of Health and…
On Nov. 19, 2020, Novartis announced that it had entered into an exclusive worldwide license and collaboration agreement…
On Nov. 19, 2020, Medigen Vaccine Biologics announced that preclinical results of their COVID-19 vaccine candidate had been…

On Nov. 19, 2020, Dallas-based company, Worlds Inc., the U.S. Air Force and Texas A&M University announced a…
On Nov. 19, 2020, University of Oxford researchers announced that research into the HIV-1 virus had shed light…

On Nov. 19, 2020, Eli Lilly and Incyte announced that the FDA had issued an Emergency Use Authorization…

On Nov. 18, 2020, Oregon Health & Science University (OHSU) announced an initiative that will attempt to discern…

On Nov. 18, 2020, RELIEF THERAPEUTICS announced the appointment of Syneos Healthᆴ, a leading global clinical research organization…

On Nov. 18, 2020, Todos Medical announced positive clinical proof of concept data from its lab-based rapid SARS-CoV-2…

On Nov. 18, 2020, Gilead Sciences announced topline results from the Phase 2/3 CAPELLA trial evaluating the company’s…

On Nov. 18, 2020, Pfizer and BioNTech announced that, after conducting the final efficacy analysis in their ongoing…
On Nov. 18, 2020, marks the end of the 11th Ebola outbreak in the Democratic Republic of the…

On Nov. 18, 2020, BioVersys announced a milestone by entering clinical development with the start of Phase 1…

On Nov. 17, 2020, the FDA issued an emergency use authorization (EUA) for the first COVID-19 diagnostic test…

On Nov. 17, 2020, RedHill Biopharma announced that the U.S. Food and Drug Administration (FDA) had cleared the…

On Nov. 17, 2020, the U.S. Dept. of Veterans Affairs (VA) announced it was working with the Centers…

On Nov. 17, 2020, the Mayo Clinic reported that more than 900 employees had contracted COVID-19 in the…

On Nov. 17, 2020, UT Southwestern researchers reported that being younger doesnメt protect against the dangers of COVID-19…

On Nov. 17, 2020, Moderna announced a supply agreement with the government of the United Kingdom (UK) to…

On Nov. 17, 2020, the proposed settlements between the federal government and OxyContin maker Purdue Pharma and its…

On Nov. 17, 2020, Celdara Medical announced that the National Institute of Allergy and Infectious Disease (NIAID) of…

On Nov. 17, 2020, the NIH announced funding of new research examining racial and ethnic disparities in pregnancy-related…

On Nov. 16, 2020, RedHill Biopharma announced that the U.S. Phase 2 study with opaganib (Yeliva, ABC294640)1 in…

On Nov. 16, 2020, Bausch Health announced the results of new investigational in vitro data showing that two…