FDA authorized long-acting monoclonal antibodies for pre-exposure prevention of COVID-19 in certain individuals
On Dec. 8, 2021, the U.S. Food and Drug Administration issued an emergency use authorization for AstraZenecaメs Evusheld…

On Dec. 8, 2021, the U.S. Food and Drug Administration issued an emergency use authorization for AstraZenecaメs Evusheld…

On Dec. 7, 2021, Arbutus Biopharma, X-Chem, and Proteros announced that Arbutus had identified several molecules that inhibit…

On Dec. 7, 2021, Rockefeller University scientists announced a study had demonstrated the therapeutic potential of an unusual…

On Dec. 5, 2021, Roche announced that its planned to launch the SARS-CoV-2 & Flu A/B Rapid Antigen…

On Dec. 1, 2021, Fulgent Genetics confirmed that the Companyメs RT-PCR test for SARS-CoV-2, the virus that causes…
On Nov. 29, 2021, Hologic announced that its three SARS-CoV-2 tests all detect the recently emerged Omicron variant of…

On Nov. 16, 2021, the University of Oxfordメs and AstraZeneca announced that two billion doses of the ChAdOx1…

On Nov. 11, 2021, the University of Oxford began recruiting for a Phase I trial to test an…

On Nov. 10, 2021, Meridian Bioscience announced that their Revogeneᆴ SARS-CoV-2 assay was granted Emergency Use Authorization by…

On Nov. 10, 2021, a study by researchers at Georgia State University showed that the politicization of science…

On Nov. 5, 2021, the USDA’s (USDA) National Veterinary Services Laboratories announced confirmation of SARS-CoV-2 in two spotted…

On Nov. 5, 2021, scientists at the National Institutes of Health announced they had found that a process…

On Nov. 1, 2021, for the first time in the U.S., the transmission of COVID-19 from pet parent…
On Oct. 29, 2021, research led by scientists at the National Institutes of Health announced they had identified…
On Oct. 27, 2021, Hologic announced that its Aptima SARS-CoV-2/Flu Assay was available for the simultaneous detection and…

On Oct. 27, 2021, Fulgent Genetics announced that it had launched an antibody test for COVID-19 which specifically…
On Oct. 22, 2021, the Department of Health and Human Services (HHS), announced that it had invested more…

On Oct. 21, 2021, the National Institutes of Health announced that a booster dose of the mRNA-1273 COVID-19…

On Oct. 14, 2021, the U.S. Department of Agriculture’s Agriculture Research Service (ARS) announced confirmation of SARS-CoV-2 (the…

On Oct. 14, 2021, The NIH Rapid Acceleration of Diagnostics (RADx) initiative announced that it has issued contract…

On Oct. 14, 2021, Cepheid announced it had received the CE mark for Xpert Xpress CoV-2/Flu/RSV plus, a…

On Oct. 13, 2021, the World Health Organization (WHO) honoured the late Henrietta Lacks with a WHO Director-General’s…

On Oct. 11, 2021, scientists at Washington University School of Medicine in St. Louis announced they had developed…

On Oct. 6, 2021, the U.S. Department of Agriculture’s Agriculture Research Service (ARS) announced confirmation of SARS-CoV-2 (the…

On Oct. 4, 2021, the the Secretary of Defense directed the mandatory vaccination of Service members against the…

On Sept. 29, 2021, Cepheid announced it had received Emergency Use Authorization from the U.S. Food & Drug…
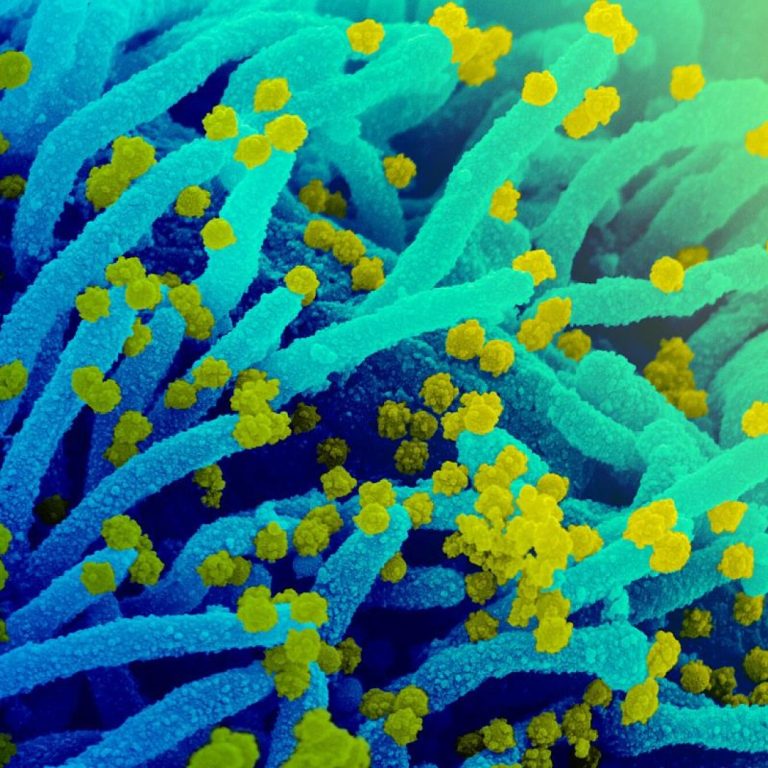
On Sept. 28, 2021, the National Institute of Allergy and Infectious Diseases announced it had awarded approximately $36.3…

On Sept. 24, 2021, the U.S. Department of Agriculture’s (USDA) announced confirmation of SARS-CoV-2 in a ferret in…

On Sept. 23, 2021, Twist Bioscience reported that its internally-discovered antibody candidate TB202-3 (CoVIC-094), demonstrated potent binding to…

On Sept. 23, 2021, Tonix Pharmaceuticals announced it had expanded its research collaboration with Columbia University. The research…