NIH announced antiviral drug development awards
On May 18, 2022, the U.S. National Institutes of Health announced it had awarded approximately $577 million to…

On May 18, 2022, the U.S. National Institutes of Health announced it had awarded approximately $577 million to…
On May 18, 2022, LabCorp announced the receipt of Emergency Use Authorization (EUA) from the U.S. Food and…
On May 17, 2022, OraSure Technologies announced that its InteliSwabᆴ COVID-19 rapid tests detect the Omicron BA.2, BA.2.12.1,…

On May 12, 2022, Cepheid announced it had received Emergency Use Authorization from the U.S. Food & Drug…

On May 6, 2022, the National Institute of Allergy and Infectious Diseases (NIAID) announced that it had launched…

On May 6, 2022, the U.S. Department of Agriculture’s Animal and Plant Health Inspection Service (APHIS) confirmed the…
On Apr. 28, 2022, the Ministry of Health of the Democratic Republic of the Congo declared an outbreak…

On Apr. 27, 2022, the U.S. Department of Agricultureメs Animal and Plant Health Inspection Service announced it had…
On Mar. 2, 2022, Sorrento Therapeutics announced that new preclinical results demonstrated that pretreating an animal with intranasal…

On Feb. 17, 2022, Novavax announced that Health Canada had granted authorization for Nuvaxovid COVID-19 Vaccine (Recombinant protein,…

On Feb. 14, 2022, The U.S. Department of Agriculture’s (USDA) Animal and Plant Health Inspective Service (APHIS) announced…

On Feb. 10, 2022, Vir Biotech announced that preclinical data suggested that sotrovimab, an investigational monoclonal antibody authorized…
On Feb. 10, 2022, Scripps Research announced that the COVID-causing virus SARS-CoV-2 harbors a vulnerable site at the…

On Feb. 4, 2022, the NIH announced a study that showed pregnant women with COVID-19 appeared to be…

On Jan. 27, 2022, Cocrystal Pharma announced that it had selected two investigational novel antiviral drug candidates for further…
On Jan. 27, 2022, NIAID announced that a clinical trial found that the combination of remdesivir plus a…

On Jan. 25. 2022, a report published in PNAS provided strong evidence of extensive SARS-CoV-2 infection of white-tailed…

On Jan. 24, 2022, the University of Oxford announced that a third booster dose of either the ChAdOx1…

On Jan. 24, 2022, Anixa Biosciences announced that the company and its partner, MolGenie, had synthesized a compound…
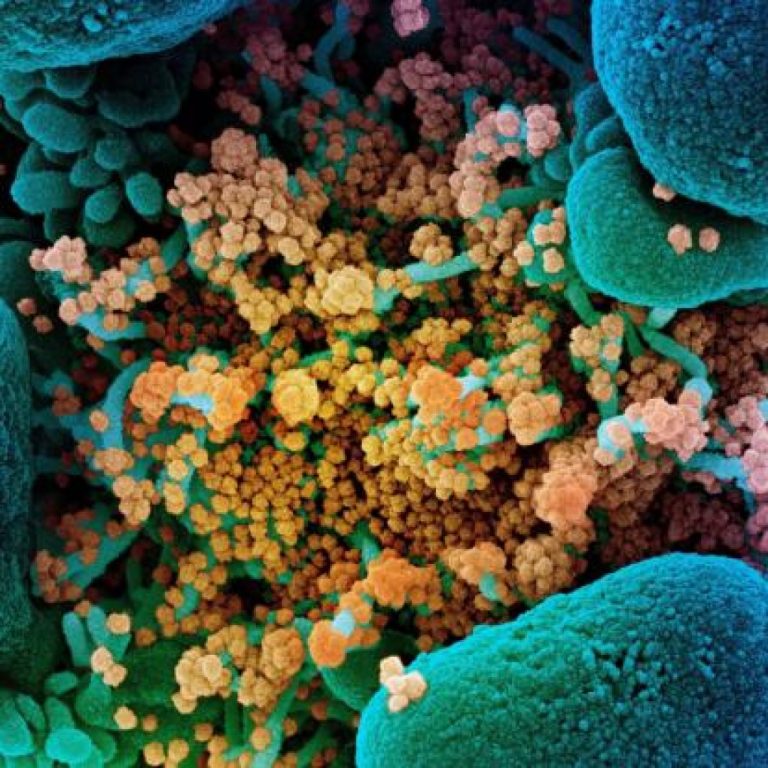
On Jan. 20, 2022, scientists from the National Institute of Allergy and Infectious Diseases (NIAID) announced that they…

On Jan. 19, 2022, Novavaxa announced that Australia’s Therapeutic Goods Administration had granted approval for provisional registration of…

On Jan. 14, 2022, the U.S. Department of Agriculture’s (USDA) National Veterinary Services Laboratories confirmed a highly pathogenic…
On Jan. 13, 2022, the Centers for Disease Control and Prevention announced a study published in the journal…
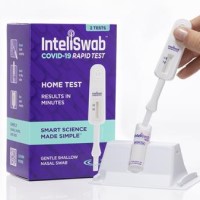
On Jan. 13, 2022, OraSure Technologies that its InteliSwabᆴ COVID-19 rapid tests detected the Omicron variant as effectively…
On Jan. 5, 2022, Pfizer and BioNTech announced a new research, development and commercialization collaboration to develop a…

On Jan 5, 2022, Hologic the addition of the Aptimaᆴ SARS-CoV-2 assay to its Global Access Initiative, a…

On Jan 5, 2022, Revelar Biotherapeutics and Twist Bioscience announced that RBT-0813 binded to and neutralized the Omicron…
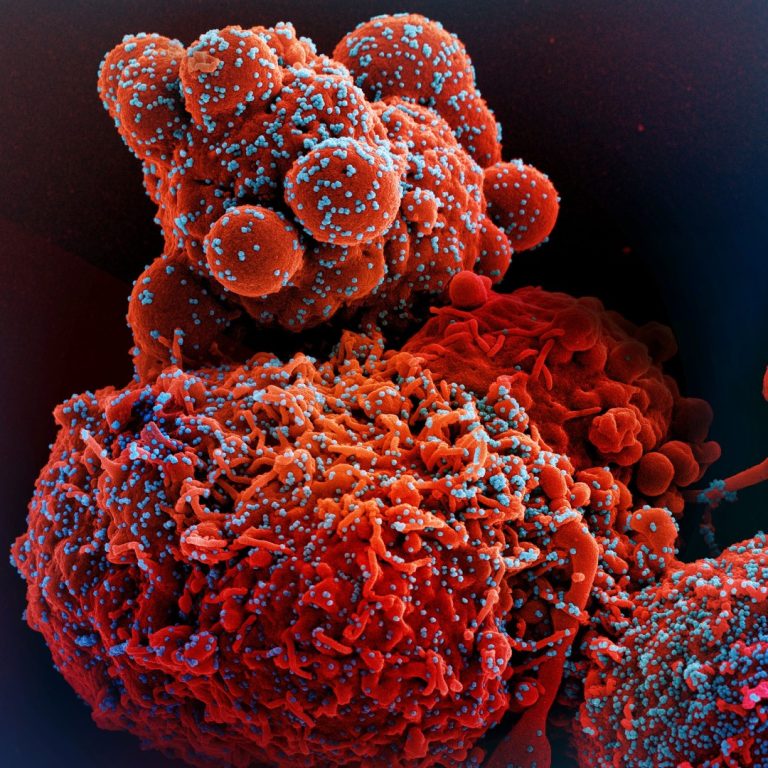
On Dec. 28, 2021, Pacific Biosciences announced that its new HiFiViral SARS-CoV-2 solution had successfully sequenced and captured…
On Dec. 28, 2021, researchers at the Walter Reed Army Institute of Research in Silver Spring, Maryland announced…
On Dec. 27, 2021, Sorrento Therapeutics announced that initial testing of COVISTIX on recombinant N proteins demonstrated its…