Uganda declared end of Ebola disease outbreak
On Jan. 11, 2023, Uganda declared the end of the Ebola disease outbreak caused by Sudan ebolavirus, less…
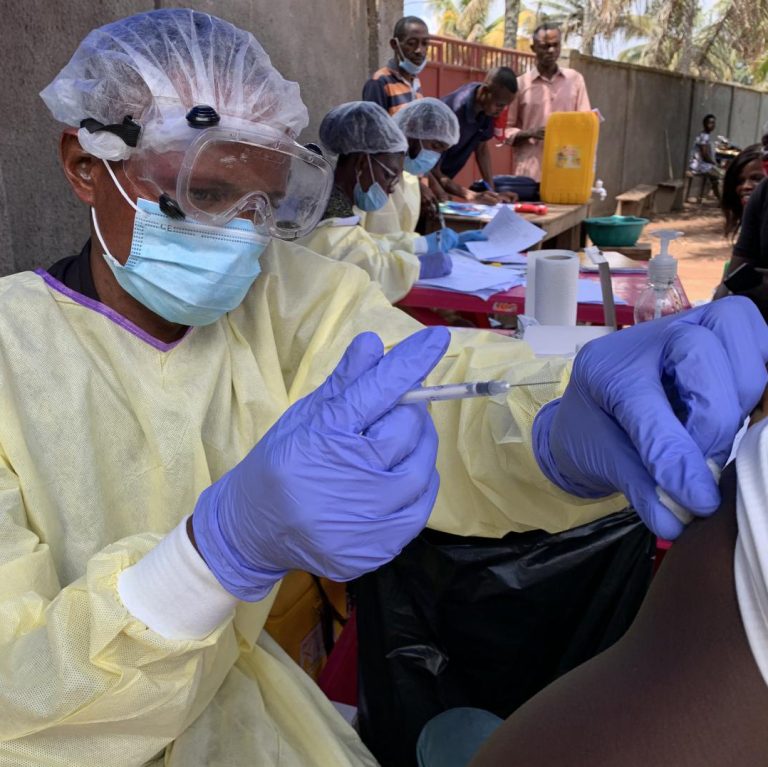
On Jan. 11, 2023, Uganda declared the end of the Ebola disease outbreak caused by Sudan ebolavirus, less…

On Jan. 11, 2023, the U.S. Food and Drug Administration reported that a single booster dose with an…

On Jan. 10, 2023, the WHO announced that an estimated 5 million children died beforecoronavirus_omicron_illustrationeir fifth birthday and…
On Jan. 9, 2023, BD (Becton, Dickinson) and CerTest Biotec have announced Emergency Use Authorization (EUA) from the…

On Jan. 4, 2023, a National Institutes of Health funded study reported that moderate levels of two outdoor…

On Dec. 20, 2022, the University of Oxford announced study results using data from 7 million people notified…
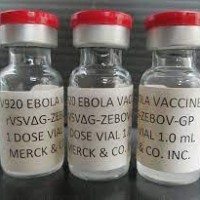
On Dec. 14, 2022, the National Institutes of Health announced that two randomized, placebo-controlled trials evaluating three Ebola…

On Dec. 8, 2022, Moderna announced it had received emergency use authorization from the U.S. Food and Drug…

On Dec. 7, 2022, Novavax announced that Health Canada had approved a supplement to a New Drug Submission…
On Dec. 7, 2022, Pfizer announced that the U.S. Food and Drug Administration (FDA) had accepted for priority…

On Nov. 22, 2022, the U.S. Food and Drug Administration approved the investigational gene therapy etranacogene dezaparvovec or…

On Nov. 21, 2022, the National Institutes of Health awarded more than $12 million to three institutions for…

On Nov. 18, 2022, Novavax announced that Health Canada had granted expanded authorization for Nuvaxovid (COVID-19 Vaccine (Recombinant…
On Nov. 15, 2022, Roche announced that the U.S. Food and Drug Administration had granted Emergency Use Authorization…

On Nov. 9, 2022, researchers at the University of Missouri announced they had identified the highly prevalent, specific…
On Nov. 9, 2022, Novavax announced that the Medicines and Healthcare products Regulatory Agency (MHRA) in the United…

On Nov. 3, 2022, Health Canada authorized an adapted version of the Moderna Spikevax COVID-19 vaccine that targets…

On Nov. 2, 2022, AIM ImmunoTech announced that the U.S. Food and Drug Administration had granted Orphan Drug…
On Oct. 25, 2022, Pacific Northwest National Laboratory (PNNL) announced that scientists have shown that they can detect…
On Oct. 20, 2022, the National Institutes of Health announced that a three-dose course of the hepatitis B…

On Oct. 19, 2022, Moderna announced that the European Medicines Agency’s (EMA) Committee for Medicinal Products for Human…
On Oct. 12, 2022, Moderna announced that it had received emergency use authorization from the U.S. Food and…

On Oct. 11, 2022, researchers from the University of Oxford have reported new findings from a Phase 1…

On Oct. 10, 2022, Novavax announced that partner, SK bioscience, had submitted a Post Approval Change Application to…

On Oct. 10, 2022, Novavax announced that Switzerland’s Federal Office of Public Health had recommended Nuvaxovid (NVX-CoV2373) as…

On Oct. 4, 2022, the United States Department of Agriculture’s (USDA) Agricultural Research Service (ARS), Southeast Poultry Research…
On Oct. 4, 2022, Washington University in St. Louis announced it had licensed the rights to develop, manufacture…
On Sept. 20, 2022, The health authorities in Uganda declared an outbreak of Ebola after a case of…

On Sept. 16, 2022, Novavax announced that the Taiwan Food and Drug Administration had granted expanded emergency use…

On Sept. 16, 2022, Novavax announced that the Israel Ministry of Health had granted an import and use…