Meridian Bioscience received FDA Emergency Use Authorization for Revogeneᆴ SARS-CoV-2 molecular assay
On Nov. 10, 2021, Meridian Bioscience announced that their Revogeneᆴ SARS-CoV-2 assay was granted Emergency Use Authorization by…

On Nov. 10, 2021, Meridian Bioscience announced that their Revogeneᆴ SARS-CoV-2 assay was granted Emergency Use Authorization by…

On Nov. 9, 2021, Moderna announced that it has submitted for a variation to the conditional marketing authorization…

On Nov. 8, 2021, Cocrystal Pharma announced that its SARS-CoV-2 main protease inhibitors showed potent in vitro pan-viral…

On Nov. 5, 2021, Chugai Pharmaceutical, announced that it had obtained approval from the Ministry of Health, Labour…

On Nov. 5, 2021, the USDA’s (USDA) National Veterinary Services Laboratories announced confirmation of SARS-CoV-2 in two spotted…

On Nov. 5, 2021, scientists at the National Institutes of Health announced they had found that a process…

On Nov. 5, 2021, Pfizer announced it was investigational novel COVID-19 oral antiviral candidate, PAXLOVID, significantly reduced hospitalization…

On Nov. 3, 2021, the World Health Organization (WHO) issued an emergency use listing (EUL) for COVAXINᆴ (developed…
On Nov. 2, 2021, the National Institutes of Health announced support for a four-year follow-up study on the…

On Nov. 1, 2021, the U.S. Food and Drug Administration (FDA) cleared the first 510(k) for a COVID-19…
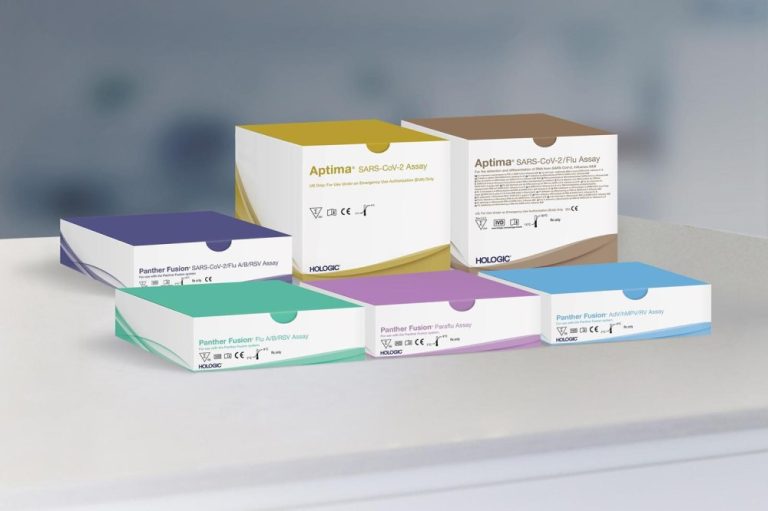
On Oct. 27, 2021, Hologic announced that its Aptima SARS-CoV-2/Flu Assay was available for the simultaneous detection and…

On Oct. 27, 2021, RELIEF THERAPEUTICS announced that its wholly owned subsidiary, APR Applied Pharma Research, reported positive…

On Oct. 21, 2021, the National Institutes of Health announced that a booster dose of the mRNA-1273 COVID-19…

On Oct. 20, 2021, Moderna announced that the U.S. Food and Drug Administration (FDA) had authorized for emergency…

On Oct. 19, 2021, National Science Foundation funded scientists at the Yale School of Public Health and the…

On Oct. 15, 2021, Pfizer and BioNTech announced they had submitted data supporting the vaccination of children 5…

On Oct. 14, 2021, The NIH Rapid Acceleration of Diagnostics (RADx) initiative announced that it has issued contract…

On Oct. 14, 2021, Cepheid announced it had received the CE mark for Xpert Xpress CoV-2/Flu/RSV plus, a…

On Oct. 14, 2021, the U.S. Department of Agriculture’s Agriculture Research Service (ARS) announced confirmation of SARS-CoV-2 (the…
On Oct. 7, 2021, PerkinElmer announced that the U.S. Food and Drug Administration (FDA) had issued Emergency Use…

On Oct. 7, 2021, Vaxart announced that a Duke University-led study published in bioRxiv showed that Vaxart’s (investigational…

On Oct. 6, 2021, the U.S. Department of Agriculture’s Agriculture Research Service (ARS) announced confirmation of SARS-CoV-2 (the…

On Oct. 5, 2021, PerkinElmer announced that the U.S. Food and Drug Administration had provided Emergency Use Authorization…
On Sept. 30, 2021, Trevena announced data from 30 patients enrolled in the proof-of-concept study of TRV027, the…

On Sept. 29, 2021, Cepheid announced it had received Emergency Use Authorization from the U.S. Food & Drug…

On Sept. 29, 2021, VBI Vaccines announced positive results from multiple preclinical studies against several COVID-19 variants of…
On Sept. 28, 2021, the National Institute of Allergy and Infectious Diseases announced it had awarded approximately $36.3…

On Sept. 28, 2021, AIM ImmunoTech announced that is had submitted a Pre-Investigational New Drug application (Pre-IND) to…

On Sept. 28, 2021, Soligenix announced publication of pre-clinical immunogenicity studies for CiVax (heat stable COVID-19 vaccine program)…

On Sept. 27, 2021, Pfizer announced the start of the Phase 2/3 EPIC-PEP (Evaluation of Protease Inhibition for…