St. Judes Reports Promising results from first prenatal therapy for spinal muscular atrophy
On Feb. 19, 2025, scientists at St. Jude Children’s Research Hospital report results from a promising new approach…
On Feb. 19, 2025, scientists at St. Jude Children’s Research Hospital report results from a promising new approach…

On Feb. 13, 2025, Ralph L. Abraham, MD, the Louisiana Surgeon General announced that the state will no…

On Jan. 30, 2025, the California Institute for Regenerative Medicine (CIRM) announced it has awarded nearly $100 million…

On Dec. 20, 2024, Novo Nordisk announced that the U.S. Food and Drug Administration (FDA) had approved Alhemo®…

On Dec. 16, 2024, a study published in BMC Cardiovascular Disorders shows that pediatric and young adult COVID-19…
On Nov. 21, 2024, the California Institute for Regenerative Medicine (CIRM) announced it had approved awarding $29.7 million to…

On Nov. 14, 2024, the U.S. Food and Drug Administration approved PTC Therapeutics’ Kebilidi (eladocagene exuparvovec-tneq), an adeno-associated…
On Oct. 11, 2024, Pfizer announced that the U.S. Food and Drug Administration (FDA) had approved HYMPAVZI™ (marstacimab-hncq)…
On Aug. 6, 2024, the U.S. Food and Drug Administration (FDA) announced it had approved vorasidenib (Voranigo, Servier…

On Apr. 3, 2024, the U.S. Food and Drug Administration approved Basilea Pharmaceutica’s Zevtera (ceftobiprole medocaril sodium for…

On Mar. 12, 2024, the United Nations Inter-agency Group for Child Mortality Estimation (UN IGME) reported that the…

On Dec. 18, 2023, the U.S. Centers for Disease Control and Prevention (CDC) announced a study that showed…

On Nov. 16. 2023, the U.S. Centers for Disease Control and Prevention (CDC) announced that Following years of…
On Nov. 9, 2023, the U.S. Food and Drug Administration approved ‘ Takeda PharmaceuticalsVAdzynma, the first recombinant (genetically…

On Oct. 20, 2023, the U.S. Centers for Disease Control and Prevention (CDC) reported that among children and…

On Oct. 14, 2023, Purdue University ensured that the memory of the former graduate and devoted Boilermaker football…

On Jun. 22, 2023, the U.S. Food and Drug Administration (FDA) approved Sarepta Therapeutics’s Elevidys, the first gene…

On Jun. 20, 2023, the U.S. Food and Drug Administration (FDA) approved Boehringer Ingelheim’s Jardiance (empagliflozin) and Synjardy…

On Apr. 27, 2023, Pfizer announced that the U.S. Food and Drug Administration (FDA) has approved PREVNAR 20®…
On Apr. 17, 2023, the U.S. Food and Drug Administration (FDA) approved Gamida Cell’s Omisirge (omidubicel-onlv), a substantially…

On Apr. 5, 2023, Pfizer announced that experimental respiratory syncytial virus (RSV) vaccine was 82% effective in preventing…
On Mar. 14, 2023, the U.S. Food and Drug Administration (FDA) amended the emergency use authorization (EUA) of…
On Mar. 8, 2023, Amphastar Pharmaceuticals announced that the U.S. Food and Drug Administration (FDA) had granted approval…

On Mar. 1, 2023, BioNTech and Pfizer announced that they had submitted an application to the U.S. Food…

On Dec. 16, 2022, Moderna announced that the European Medicines Agency’s Committee for Medicinal Products for Human Use…
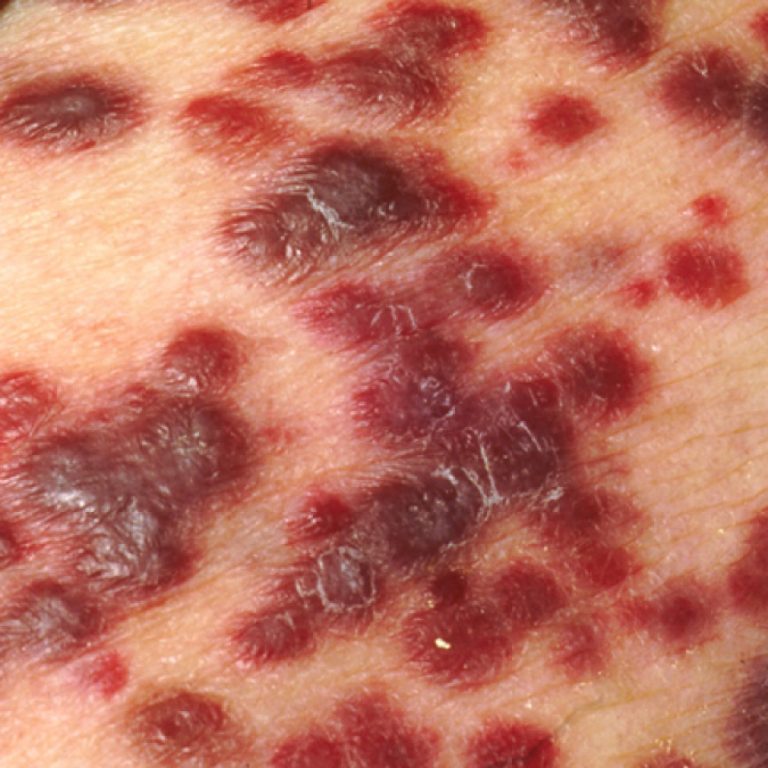
On Dec. 9, 2022, the U.S. Food and Drug Administration (FDA) approved Genentech’s atezolizumab (Tecentriq) for adult and…

On Dec. 5, 2022, Pfizer and BioNTech announced that the companies had submitted an application to the U.S….

On Nov. 17, 2022, the U.S. Food and Drug Administration (FDA) approved Tzield (teplizumab-mzwv) injection to delay the…

On Aug. 17, 2022, the U.S. Food and Drug Administration approved Zynteglo (betibeglogene autotemcel) developed by bluebird bio,…

On Jun. 23 2022, Novavax announced the filing of a Supplement to a New Drug Submission with Health…