Study Finds Declining Perceptions of Safety of Covid-19, Flu, and MMR Vaccines
On Feb. 10, 2026, an Annenberg survey finds that a sizable majority of Americans think the three vaccines…

On Feb. 10, 2026, an Annenberg survey finds that a sizable majority of Americans think the three vaccines…
On May 12, 2025, the the Ministry of Health (MoH) of the Kingdom of Saudi Arabia (KSA) reported…
On May 1, 2025, the U.S. Centers for Disease Control and Prevention (CDC) announced that hospitals are no…

On Apr. 15, 2025, Moderna announced that its manufacturing facility in Harwell, Oxfordshire, the Moderna Innovation and Technology…

On Apr. 15, 2025, Novavax announced preliminary results from the SHIELD-Utah study (Study of Healthcare Workers and First…

On Apr. 4, 2025, the World Health Organization (WHO) announced that over the past two days, it convened…
On Apr. 30, 2024, a meta-analysis of six randomized controlled trials in JAMA Neurology found no increase in seizures in…
On Apr. 12, 2024, Oregon Health Sciences University (OHSU) announced research that shows using live SARS-CoV-2 virus revealed…

On Mar. 22, 2024, the U.S. Food and Drug Administration (FDA) issued the an emergency use authorization for…
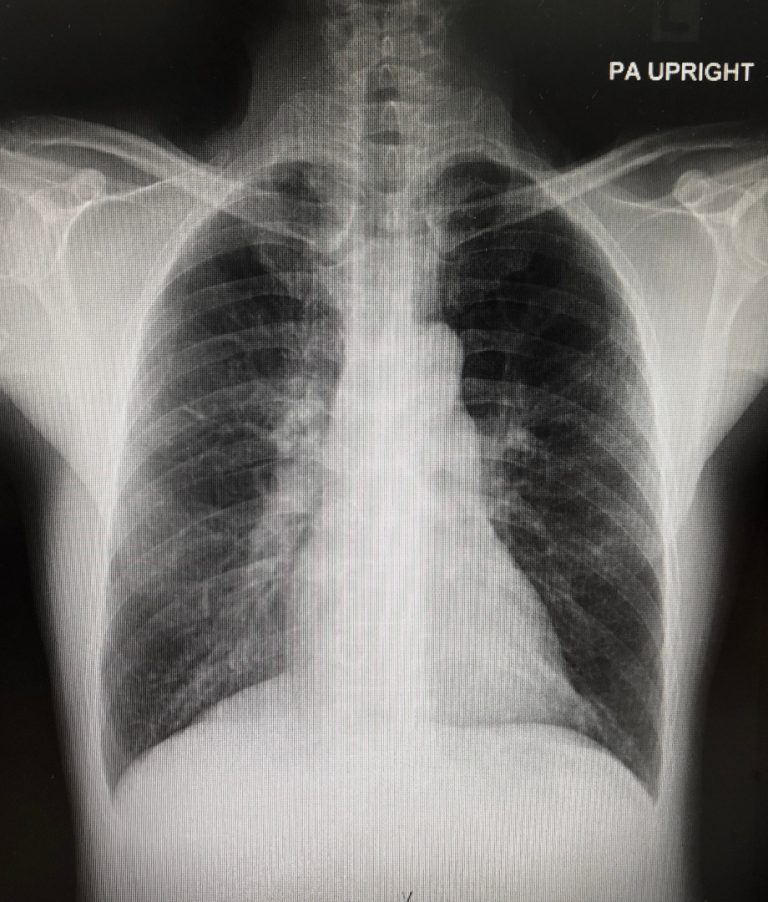
On Mar. 20, 2024, a National Institutes of Health-supported study reported that SARS-CoV-2, the virus that causes COVID-19,…
On Apr. 23, 2023, the most comprehensive state-by-state analysis of the impacts of COVID-19 across the USA, published…

On Apr. 4, 2023, NanoViricides announced that it had executed a license agreement with Karveer Meditech, Kolhapur, India,…

On Mar. 21, 2023, Quest Diagnostics announced that it had introduced two innovative Post-COVID-19 panels. These laboratory tests…

On Mar. 20, 2023, National Institutes of Health (NIH) scientists announced that the magnitude and quality of a…
On Mar. 14, 2023, the U.S. Food and Drug Administration (FDA) amended the emergency use authorization (EUA) of…

On Mar. 1, 2023, BioNTech and Pfizer announced that they had submitted an application to the U.S. Food…

On Dec. 5, 2022, Pfizer and BioNTech announced that the companies had submitted an application to the U.S….
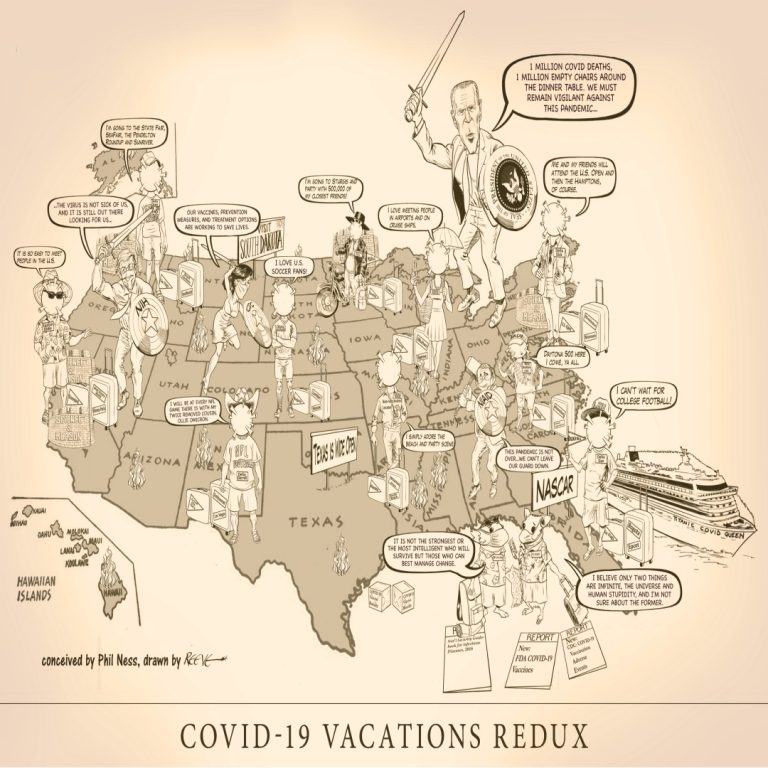
COVID-19 Vacations Redux iIlustrates once again the pits and perils vacation travelers face as they begin their Summer…
Play the COVID-19 Wheel of Fortune and see how lucky you are! You have two wheels to choose…

On Apr. 27, 2022, the U.S. Department of Agriculture’s Animal and Plant Health Inspection Service announced it had…

On Apr. 22, 2022, the World Health Organization (WHO) made a strong recommendation for nirmatrelvir and ritonavir, sold…

On Apr. 20, 2022, Codiak BioSciences announced announc a Non-Clinical Evaluation Agreement (NCEA) with the National Institute of…

On Apr. 19, 2022, Novavax announced that its partner, Takeda, received manufacturing and marketing approval from the Japan…

On Apr. 18, 2022, the U.S. Food and Drug Administration (FDA) amended the emergency use authorizations (EUAs) of…

On Mar. 14, 2022, Pfizer and BioNTech announced positive results from a Phase 2/3 clinical trial evaluating the…

On Apr. 14, 2022, the U.S. Food and Drug Administration issued an emergency use authorization (EUA) for the…
On Apr. 14, 2022, Merck announced that V116, the companyメs investigational 21-valent pneumococcal conjugate vaccine, had received Breakthrough…

On Apr. 13, 2022, Novavax announced that Swissmedic had granted Novavax conditional marketing authorization (CMA) for Nuvaxovid COVID-19…
On Apr. 11, 2022, Veru announced positive efficacy and safety results from a planned interim analysis of the…

On Apr. 8, 2022, Novavax announced and Serum Institute of India (SII), the world’s largest vaccine manufacturer by…