The FDA announced actions to accelerate development of novel prevention, treatment options for COVID-19
On May 11, 2020, the FDA announced actions to accelerate the development of prevention and treatment options for…

On May 11, 2020, the FDA announced actions to accelerate the development of prevention and treatment options for…

On May 8, 2020, the NIH announced that a randomized, controlled clinical trial evaluating the safety and efficacy…

On May 7, 2020, Catalent announced Phase 3 study for lenzilumab, the companyメs proprietary Humaneeredᆴ anti-human granulocyte macrophage-colony…

On May 6, 2020, Predictive Oncology announced the acquisition of Soluble Therapeutics and partnership and licensing of a…

On May 5, 2020, Organicell Regenerative Medicine announced the FDA has approved the Investigational New Drug application for…

On May 5, 2020, Pfizer and BioNTech announced the first participants have been dosed in the U.S. in…

On May 5, 2020, the HonorHealth Research Institute and HonorHealth announced the successful enrollment on a novel combination…

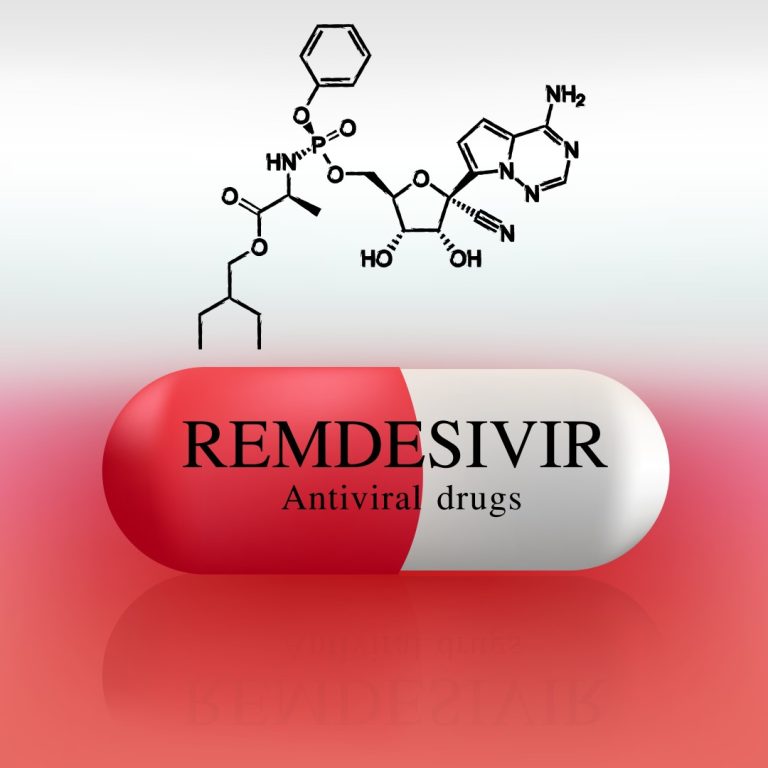
On May 1, 2020, the FDA issued an Emergency Use Authorization (EUA) for the investigational antiviral drug remdesivir…

On May 1, 2020, the U.S. Dept. of Veterans Affairs (VA) announced participation in a series of clinical…
On Apr. 29, 2020, the National Institutes of Health (NIH) announced that hospitalized patients with advanced COVID-19 and…

On Apr. 29, 2020, an anti-inflammatory drug developed at Scripps Research 25 years ago tested as a way…

On Apr. 29, 2020, Emory University played a leading role in the government-sponsored clinical trial of the COVID-19…
On Apr. 29, 2020, researchers at Montefiore Health System and Albert Einstein College of Medicine and NYU Langone…

On Apr. 29, 2020, researchers at Montefiore Health System and Albert Einstein College of Medicine announced that they…
On Apr. 29, 2020, a team of Texas A&M University researchers asked hundreds of frontline medical workers to…

On Apr. 28, 2020, INOVIO announced that its Phase 1 U.S. clinical trial for COVID-19 DNA vaccine INO-4800…

On Apr. 24, 2020, BioSig Technologies announced that subsidiary ViralClear had submitted an Investigational New Drug (IND) application…

On Apr. 23, 2020, Yale researchers began a clinical trial at Yale New Haven Hospital to test the…

On Apr. 22, 2020, BioNTech and Pfizer announced that the German regulatory authority, the Paul-Ehrlich-Institut, has approved the…

On Apr. 20, 2020, SNBL announced their intention to participate in the development of a prophylactic DNA vaccine…

On Apr. 21, 2020, Rutgers University announced it has launched the nation’s largest prospective study of health care…

On Apr. 21, 2020, Immunic reported that its lead asset, IMU-838, a selective oral DHODH inhibitor successfully demonstrated…

On Apr. 20, 2020, Novartis announced it had reached an agreement with the U.S. Food and Drug Administration…

On Apr. 20, 2020, Atossa Therapeutics announced that it had applied to the FDA for approval to commence…

On Apr. 17, 2020, the NIH and the Foundation for the NIH are bringing together more than a…

On Apr. 17, 2020, Sinovac Biotech announced that the Company had commenced Phase I clinical trial, a randomized,…

On Apr. 17, 2020, Incyte announced the initiation of RUXCOVID, a global, randomized, double-blind, placebo-controlled phase 3 clinical…

On Apr. 17, 2020, Emory University announced it had begun enrolling adults over age 55 in the clinical…

On Apr. 16, 2020, the International Vaccine Institute (IVI) announced that the Coalition for Epidemic Preparedness Innovations (CEPI)…

On Apr. 16, 2020, BioSig Technologies announced that its subsidiary ViralClear Pharmaceuticals, had updated its clinical development program…