BARDA reselected ICON as preferred partner to help strengthen U.S. emergency preparedness and response
On Oct. 19, 2020, ICON announced that it had been re-selected by the Biomedical Advanced Research and Development…

On Oct. 19, 2020, ICON announced that it had been re-selected by the Biomedical Advanced Research and Development…

On Oct. 6, 2020, the National Institutes of Health (NIH), working in collaboration with the Biomedical Advanced Research…

On Aug. 11, 2020, Moderna announced that the U.S. government had secured 100 million doses of mRNA-1273. The…
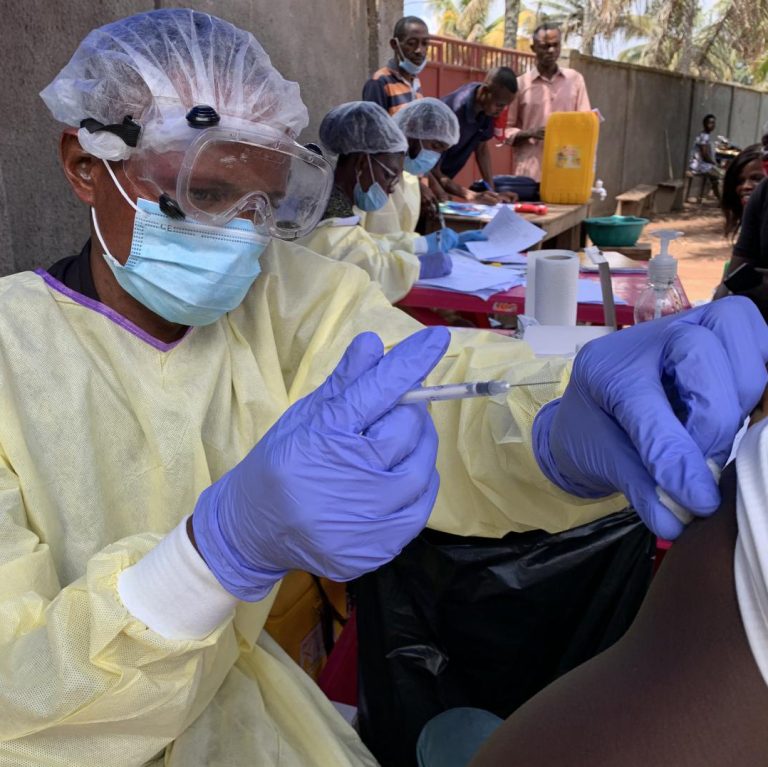
On Jul. 29, 2020, Regeneron announced that the Biomedical Advanced Research and Development Authority (BARDA) had entered into…

On Jul. 26, 2020, Moderna announced a modification to its contract with the Biomedical Advanced Research and Development…
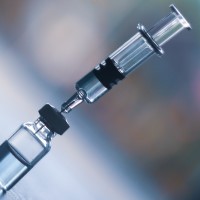
On Jul. 8, 2020, BD (Becton, Dickinson) announced the formation of a strategic, public-private partnership with the Biomedical…

On Jul. 7, 2020, Regeneron announced that, as part of Operation Warp Speed, the Biomedical Advanced Research and…

On Jun. 30, 2020, Altimmune announced dosing of the first patient in the Company’s Phase 1b clinical trial…
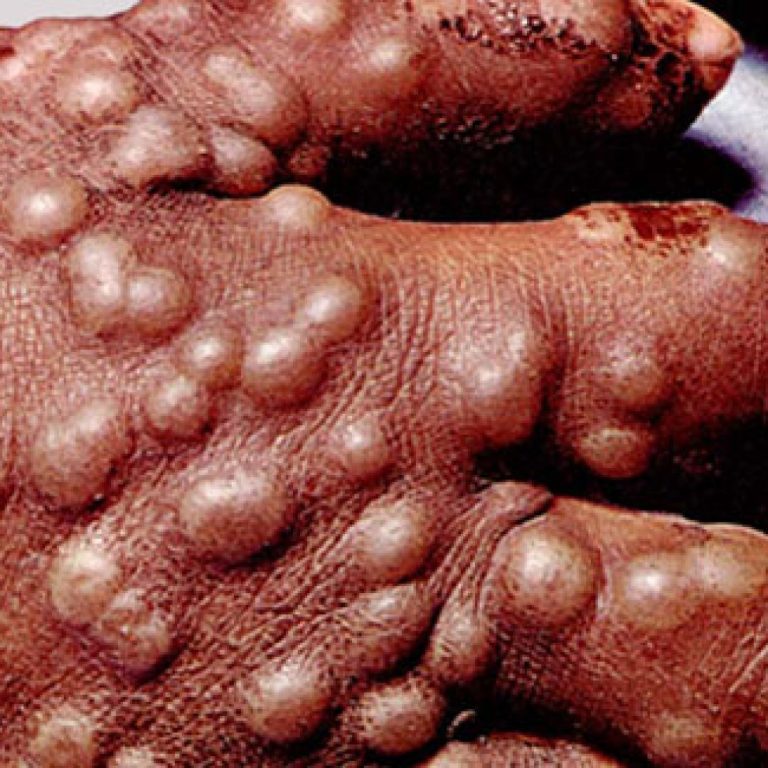
On Jun. 25, 2020, SIGA Technologies announced the deliveries of oral TPOXX (tecovirimat) to the U.S. Department of…

On Jun. 11, 2020, Quidel announced it had received funding from the Biomedical Advanced Research and Development Authority…

On Jun. 10, 2020, OraSure Technologies announced it had been awarded a $629,217 contract from the Biomedical Advanced…

On Jun. 1, 2020, Emergent BioSolutions announced it had been issued a task order under an existing contract…

On May 19, 2020, Phlow, a U.S.-based, public benefit drug manufacturing corporation, received federal government funding of $354…

On May 4, 2020, Mayo Clinic was awarded a $26 million contract from the Biomedical Advanced Research and…

On Apr. 30, 2020, InBios announced that it had been awarded a contract from the Biomedical Advanced Research…

On Apr. 27, 2020, Moderna announced it had submitted an Investigational New Drug application to the U.S. Food…

On Apr. 22, 2020, the Biomedical Advanced Research and Development Authority (BARDA) and Tangen Biosciences announced teaming up…
On Apr. 16, 2020, the U.S. government announced it will fund development of an experimental coronavirus vaccine from…
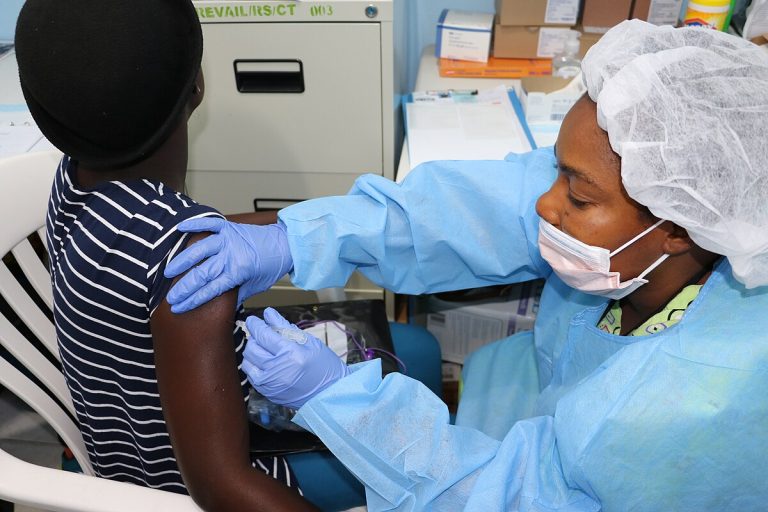
On Apr. 16, 2020, Regeneron announced the U.S. Food and Drug Administration (FDA) had accepted for Priority Review…
On Apr. 8, 2020, Nanomix announced that the company had been awarded $570,000 in funding from Biomedical Advanced…

On Apr. 6, 2020, OraSure Technologies announced it had been awarded a $710,310 contract from the Biomedical Advanced…
On Apr. 2, 2020, Emergent BioSolutions announced it had entered into a formal partnership with the U.S. government…

On Mar. 31, 2020, Philips announced that it was fully committed to fulfill the contract with the U.S….

On Mar. 31, 2020, Cue Health announced that is had been awarded up to $30 million in base…
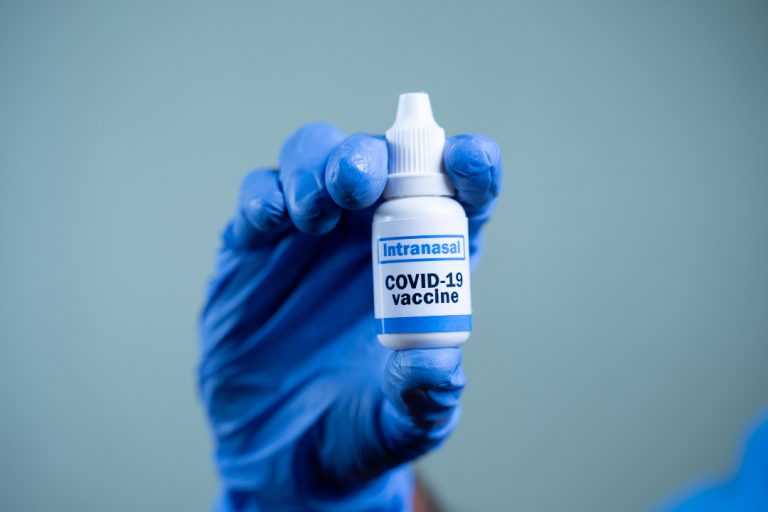
On Mar. 30, 2020, Altimmune announced that it was launching a collaboration with the University of Alabama at…
On Mar. 19, 2020, Roche announced it was working with the U.S. Food & Drug Administration (FDA) to…

On Mar. 19, 2020, Mesa Biotech announced it has significantly expanded its capacity to test clinical samples for…
On Feb. 18, 2020, Sanofi announced it had leveraged previous development work for a SARS vaccine which may…

On Jul. 12, 2018, Cue Health announced that is had been awarded up to $30 million in base…
On Jul. 13, 2010, Bavarian Nordic announced that it had delivered 1 million doses of its smallpox vaccine…