WHO launched guide on whole genome sequencing use as a tool for foodborne disease surveillance and response
On Nov. 7, 2023, the World Health Organization (WHO) announced that it had released a new guidance: モWhole…
On Nov. 7, 2023, the World Health Organization (WHO) announced that it had released a new guidance: モWhole…
On Nov. 1, 2023, the International Health Regulations (IHR) National Focal Point of Zambia notified WHO of an…

On Oct. 12, 2023, the U.S. Centers for Disease Control and Prevention (CDC) announced that two fatal human…

On Oct. 11, 2023, the European Commission, the European Investment Bank and the Bill & Melinda Gates Foundation…
On Oct. 2, 2023, the World Health Organization (WHO) announced it had recommended a new vaccine, R21/Matrix-M, developed…

On Sept. 14, 2023, the World Health Organization (WHO) reported that as of September 11, 2023, a total…

On Aug. 29, 2023, the COVID-19 Technology Access Pool (C-TAP), a multi-stakeholder partnership to facilitate sharing of intellectual…
On Aug. 9, 2023, the World Health Organization (WHO) released standing recommendations for battling COVID-19 and released an…
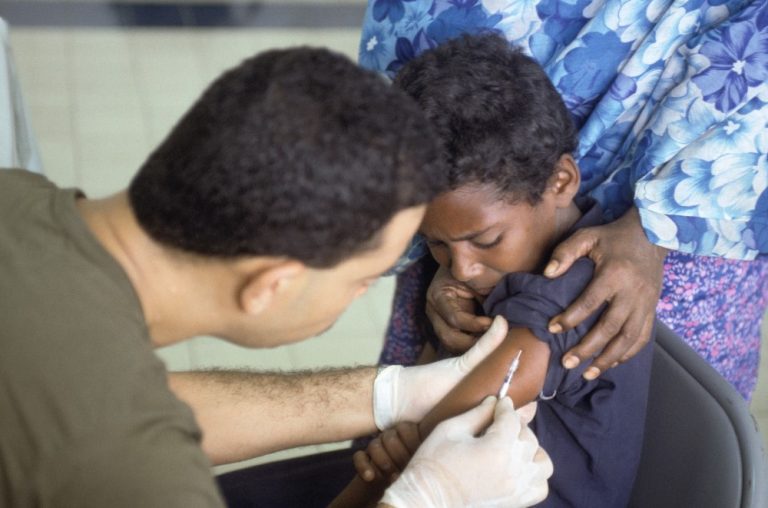
On Jul. 28, 2023, the World Health Organization (WHO) reported circulating vaccine-derived poliovirus type 2 (cVDPV2) in the…

On Jul. 27, 2023, the IHR National Focal Point of Poland notified WHO of unusual deaths in cats…
On Jul. 24, 2023, the United Arab Emirates (UAE), notified WHO of a case of Middle East Respiratory…

On Jul. 5, 2023, the IHR National Focal Point of Poland notified WHO of unusual deaths in cats…
On May 31, 2023, the World Health Organization announced it had assigned simple, easy to say and remember…

On May 30, 2023, the United Kingdom of Great Britain and Northern Ireland reported to the World Health…
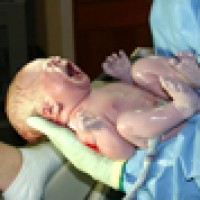
On May 9, 2023, United Nations agencies and partners reported that an estimated 13.4 million babies were born…
On May 5, 2023, the World Health Organization (WHO) ended its declaration of COVID-19 as a global health…

On Apr. 15, 2023, the World Health Organization (WHO) announced that six additional laboratory-confirmed cases of Marburg virus…

On Apr. 5, 2023, the Food and Agriculture Organization (FAO), in collaboration with the World Health Organization (WHO),…

On Mar. 29, 2023, the World Health Organization (WHO) announced that it had certified Azerbaijan and Tajikistan for…

On Mar. 17, 2023, the Indonesia Ministry of Health notified the World Health Organization (WHO) of the detection…

On Feb. 26, 2023, the Cambodia International Health Regulations (IHR) National Focal Point (NFP) reported one confirmed case…

On Feb. 3, 2023, the World Health Organization (WHO) released an updated Global Breast Cancer Initiative (GBCI) Framework…

On Jan. 10, 2023, the WHO announced that an estimated 5 million children died beforecoronavirus_omicron_illustrationeir fifth birthday and…

On Jan. 1, 2023, a study published in the Lancet reported that concentrations of antibiotic residues found in…

On Nov. 12, 2022, the World Health Organization (WHO) issued a Disease Outbreak News (DON) on the circulating…
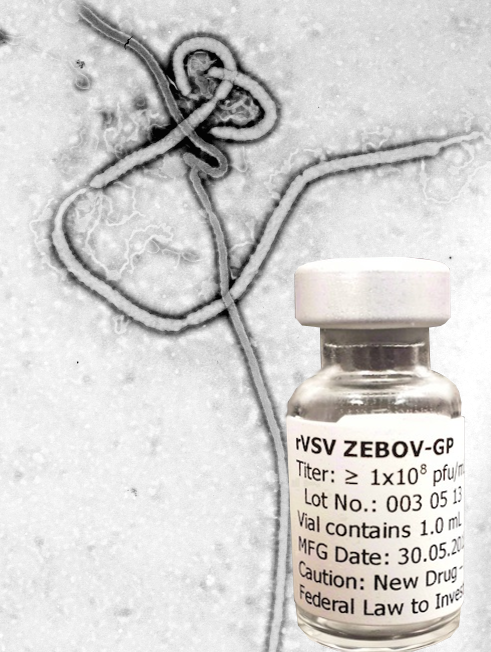
On Dec. 15, 2022, the University of Oxford’s Ebola vaccine candidate has been shipped to Uganda, just 80…

On Dec. 9, 2022, the World Health Organization (WHO) announced that the first doses of one of the…

On Dec. 7, 2022, Novavax announced that Health Canada had approved a supplement to a New Drug Submission…

On Nov. 29, 2022, Novavax announced that the World Health Organization had issued an updated Emergency Use Listing…
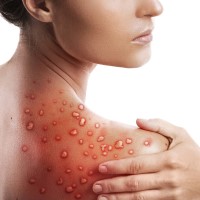
On Nov. 28, 2022, the WHO announced that following a series of consultations with global experts, it had…