Circulating vaccine-derived poliovirus type 2 (cVDPV2) reported in Indonesia
On Nov. 12, 2022, the World Health Organization (WHO) issued a Disease Outbreak News (DON) on the circulating…

On Nov. 12, 2022, the World Health Organization (WHO) issued a Disease Outbreak News (DON) on the circulating…

On Dec. 16, 2022, Moderna announced that the European Medicines Agency’s Committee for Medicinal Products for Human Use…
On Dec. 16, 2022, Novavax announced that the Standing Committee on Vaccination (STIKO) in Germany had expanded its…
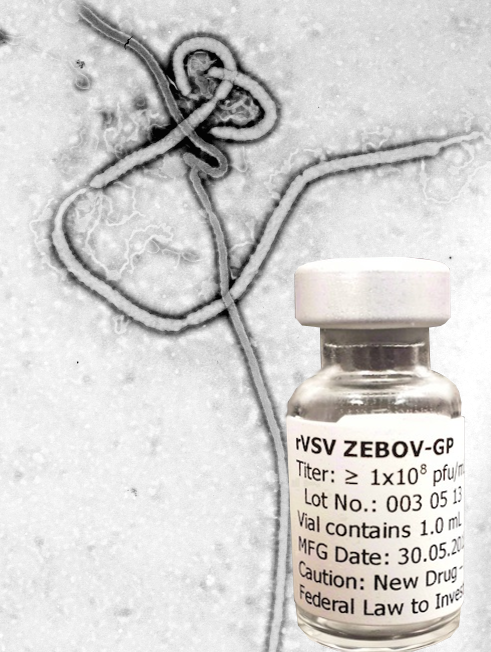
On Dec. 15, 2022, the University of Oxford’s Ebola vaccine candidate has been shipped to Uganda, just 80…
On Dec. 14, 2022, the National Institutes of Health announced that two randomized, placebo-controlled trials evaluating three Ebola…

On Dec. 12, 2022, the Oregon Department of Agriculture and the U.S. Department of Agriculture’s Animal Plant Health…

On Dec. 11, 2022, Novavax announced that the Switzerland’s Federal Office of Public Health and Francees Haute Autorite…

On Dec. 10, 2022, CSL announced data affirming the long-term durability and safety of single-infusion HEMGENIX® (etranacogene dezaparvovec-drlb)…

On Dec. 9, 2022, the World Health Organization (WHO) announced that the first doses of one of the…

On Dec. 9, 2022, Pfizer and BioNTech announced the companies had received Fast Track Designation from the U.S….
On Dec. 9, 2022, Washington University in St. Louis announced that it’s nasal COVID-19 vaccine technology had been…

On Dec. 8, 2022, Moderna announced it had received emergency use authorization from the U.S. Food and Drug…

On Dec. 8, 2022, Pfizer and BioNTech announced the U.S. Food and Drug Administration had granted Emergency Use…

On Dec. 7, 2022, Novavax announced that Health Canada had approved a supplement to a New Drug Submission…
On Dec. 5, 2022, the Fred Hutchinson Cancer Center announced that according to a new meta-analysis women were…

On Dec. 5, 2022, Pfizer and BioNTech announced that the companies had submitted an application to the U.S….

On Dec. 2, 2022, the Oregon Department of Agriculture (ODA) and the U.S. Department of Agriculture’s (USDA) Animal…

On Dec. 2, 2022, in a multi-center collaboration led by Scripps Research, scientists announced they have achieved promising…

On Nov. 29, 2022, Novavax announced that the World Health Organization had issued an updated Emergency Use Listing…

On Nov. 21, 2022, the WHO is launching a global scientific process to update the list of priority…

On Nov. 18, 2022, Pfizer and BioNTech announced results from an analysis examining the immune response induced by…

On Nov. 18, 2022, the World Health Organisation for Animal Health (WHO) announced that Peru had reported its…

On Nov. 18, 2022, Novavax announced that Health Canada had granted expanded authorization for Nuvaxovid (COVID-19 Vaccine (Recombinant…

On Nov. 16, 2022, Pfizer and BioNTech announced that the companies had initiated a Phase 1 study to…

On Nov. 16, 2022, the Cancer Prevention and Research Institute of Texas (CPRIT) approved $12 million in recruitment…

On Nov. 15, 2022, Moderna reported findings from a Phase II/III clinical trial where bivalent Omicron-targeting booster candidates,…

On Nov. 10, 2022, Pfizer and BioNTech announced a booster dose of their Omicron BA.4/BA.5-adapted bivalent COVID-19 vaccine…
On Nov. 9, 2022, researchers at the University of Missouri announced they had identified the highly prevalent, specific…
On Nov. 9, 2022, Novavax announced that the Medicines and Healthcare products Regulatory Agency (MHRA) in the United…
On Nov. 8, 2022, Novavax announced topline results from its Phase 3 Boosting Trial for the SARS-CoV-2 rS…