OPKO Health’s BioReference Laboratories launched COVID-19 antibody screening for New York City residents
On May 8, 2020, BioReference Laboratories, an OPKO Health company, announced the launch of COVID-19 antibody screening available…

On May 8, 2020, BioReference Laboratories, an OPKO Health company, announced the launch of COVID-19 antibody screening available…

On May 7, 2020, Entos Pharmaceuticals announced a partnership with Precision NanoSystems, Inc., (PNI), a global leader in…

On May 11, 2020, the Air Force Genetics Center of Excellence at Keesler Air Force Base announced it…
On May 7, 2020, Gilead announced that the Japanese Ministry of Health, Labour and Welfare (MHLW) has granted…

On May 7, 2020, a study of nearly 1,400 patients with moderate to severe COVID-19 disease at a…

On May 6, 2020, 3M and Ford announced that newly designed powered air-purifying respirators (PAPRs), developed are on…
On May 6, 2020, the U.S. Food and Drug Administration (FDA) approved Tabrecta (capmatinib) for the treatment of…

On May 6, 2020, Baxter announced results of the Fluid Response Evaluation in Sepsis Hypotension and Shock (FRESH)…
On May 6, 2020, Mesoblast announced that the first patients were dosed in the 300-patient randomized placebo-controlled Phase…

On May 6, 2020, bioMerieux announced performance validation and the upcoming launch of VIDAS® anti-SARS-CoV-2 serology tests to detect…

On May 6, 2020, Laurent Pharmaceuticals announced that it has obtained the approval from Health Canada to initiate…

On May 5, 2020, Athersys announced that the first patients have been enrolled in Athersys’ pivotal Phase 2/3…

On May 5, 2020, the U.S. Dept. of Veterans Affairs (VA) announced the Airborne Hazards and Open Burn…

On May 5, 2020, the HonorHealth Research Institute and HonorHealth announced the successful enrollment on a novel combination…

On May 5, 2020, Innovation Pharma reported it had executed executed a Material Transfer Agreement (MTA) with a…

On May 5, 2020, Athersys announced the first patients have been enrolled in Athersys’ pivotal Phase 2/3 study…

On May 4, 2020, the U.S. Patent and Trademark Office (USPTO) unveiled a new web-based intellectual property marketplace…

On May 4, 2020, Vir Biotech and Alnylam Pharma announced the selection of a development candidate for VIR-2703,…

On May 4, 2020, Anixa Biosciences and partner OntoChem, announced they had identified its first therapeutic candidate against…

On May 4, 2020, Cerus announced U.S. Food and Drug Administration (FDA) regulatory approval for manufacture of INTERCEPT…

On May 4, 2020, Junshi Biosciences and Eli Lilly announced they have entered into an agreement to co-develop therapeutic…

On May 4, 2020, Mayo Clinic was awarded a $26 million contract from the Biomedical Advanced Research and…

On May 3, 2020, the Government of Canada announced it was investing in projects that support the global…
On May 1, 2020, Gilead announced that the U.S. Food and Drug Administration (FDA) has granted emergency use…

On May 1, 2020, the U.S. Food and Drug Administration (FDA) issued an Emergency Use Authorization (EUA) for…
On May 1, 2020, a new University of Southern California (USC) study suggests that temporarily suppressing the body’s immune…

On May 1, 2020, the U.S. Dept. of Veterans Affairs (VA) announced participation in a series of clinical…
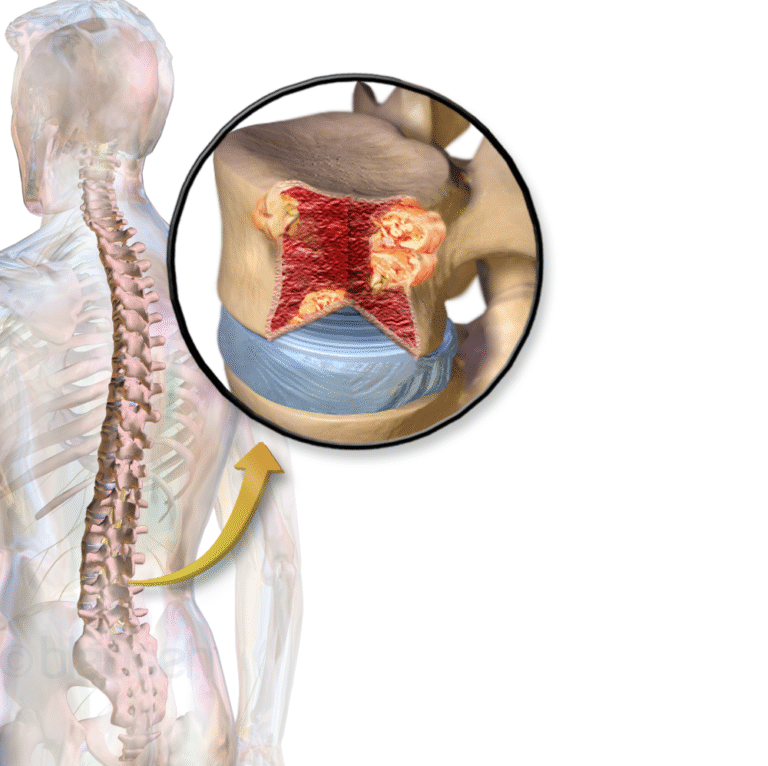
On May 1, 2020, the Food and Drug Administration (FDA) approved daratumumab and hyaluronidase-fihj (DARZALEX FASPRO, Janssen Biotech.)…

On May 1, 2020, CHF Solutions announced that it has shipped Aquadex product to a specialty care center…

On May 1, 2020, 3M announced that it has completed the sale of substantially all of its drug delivery…