Twist Bioscience collaborates with Adicet Bio to accelerate discovery of Gamma Delta T cell cancer therapeutics
On Sept. 13, 2021, Twist Bioscience announced and Adicet Bio announced a collaboration to accelerate the discovery of…

On Sept. 13, 2021, Twist Bioscience announced and Adicet Bio announced a collaboration to accelerate the discovery of…

On Sept. 9, 2021, the National Institutes of Health (NIH) announced that it was providing approximately $185 million…

On Sept. 8, 2021, National Resilience announced an agreement to manufacture mRNA for the Moderna COVID-19 vaccine. Under…

On Sept. 7, 2021, Takeda Pharmaceutical announced the Government of Japan’s Ministry of Health, Labour and Welfare purchase…

On Sept. 6, 2021, Pfizer and BioNTech announced that they had submitted a variation to the European Medicines…

On Aug. 31, 2021, Novartis announced a commercial agreement with the NHS in England as part of a…
On Aug. 30, 2021, Oragenics announced that the stabilized pre-fusion spike protein trimer produced by its Canadian collaborator…

On Aug. 30, 2021, the Mayo Clinic reported that in an observational study the combination of casirivimab and…

On Aug. 27, 2021, researchers at University of British Columbia’s (UBC) faculty of medicine and BC Cancer Research…

On Aug. 27, 2021, National Aeronautics and Space Administration (NASA) Space station astronauts successfully demonstrated DNA repair in…

On Aug. 26, 2021, Oregon Health & Science University (OHSU) announced a laboratory study found that blood serum…

On Aug. 26, 2021, Pfizer and BioNTech announced the signing of a letter of intent with Eurofarma Laboratorios,…
On Aug. 25, 2021, Anixa Biosciences announced that a genomic variant analysis indicateed that its potential compounds may…

On Aug. 25, 2021, Pfizer and BioNTech announced the initiation of a supplemental Biologics License Application (sBLA) to…

On Aug. 23, 2021, Pfizer and BioNTech announced that the U.S. Food and Drug Administration (FDA) approved the…

On Aug. 19, 2021, Oregon Health & Science University (OHSU) announced that it was part of a randomized,…

On Aug. 19, 2021, the University of Oxford reported that obtaining two vaccine doses remained the most effective…

On Aug. 17, 2021, researchers reported the delta variant of the virus that causes COVID-19 was not particularly…

On Aug. 13, 2021, Merck announced that the U.S. Food and Drug Administration (FDA) had approved WELIREG, an…

On Aug. 12, 2021, Innovation Pharma released information regarding the status of its randomized, double-blind, placebo-controlled Phase 2…

On Aug. 11, 2021, the World Health Organization (WHO) announced the next phase in its Solidarity trial that…

On Aug. 3, 2021, CytoDyn announced that Brazil’s regulatory authority ANVISA (Agência Nacional de Vigilância Sanitária) has approved…

On Jul. 29, 2021, Incyte announced the U.S. Food and Drug Administration (FDA) had broadened the Emergency Use…

On Jul. 29, 2021, BOTOX(R) Allergan, an AbbVie company, announced that the U.S. Food and Drug Administration (FDA)…
On Jul. 29, 2021, the National Institutes of Health announced it had launched a new online research tool…
On Jul. 29, 2021, SIGA Technologies announced that it had entered into a collaboration with Oxford University in…

On Jul. 29, 2021, Cocrystal Pharma announced that its SARS-CoV-2 3CL protease lead CDI-45205 and several analogs showed…
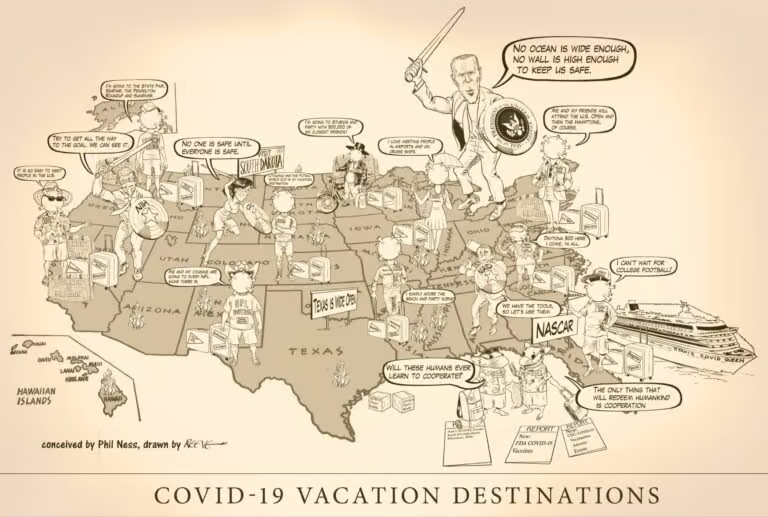
Planning a Summer vacation? Where to go? How about going to one of those large events with tens…
On Jul. 22, 2021, Moderna announced a new supply agreement with Taiwan for 20 million doses of Moderna’s…
On Jul. 16, 2021, Merck announced the U.S. Food and Drug Administration (FDA) had approved VAXNEUVANCE (Pneumococcal 15-valent…