Medical imaging breakthrough could transform cancer and arthritis diagnosis
On Sept. 30, 2024, a University College London research team announced technology that can deliver photoacoustic tomography (PAT)…
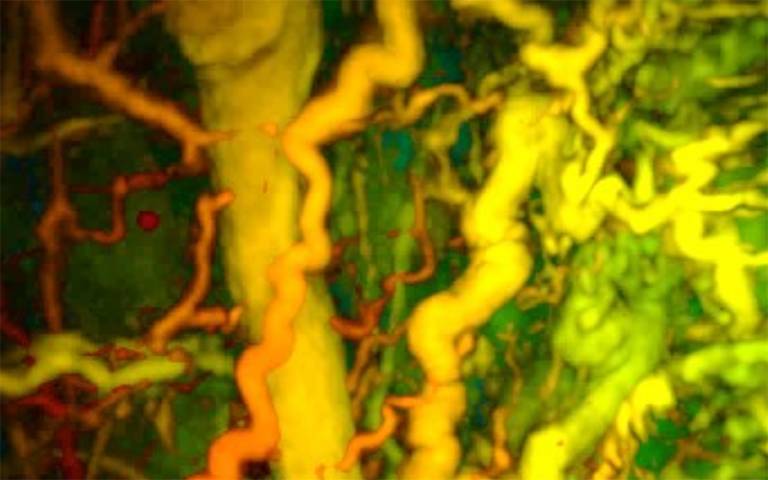
On Sept. 30, 2024, a University College London research team announced technology that can deliver photoacoustic tomography (PAT)…
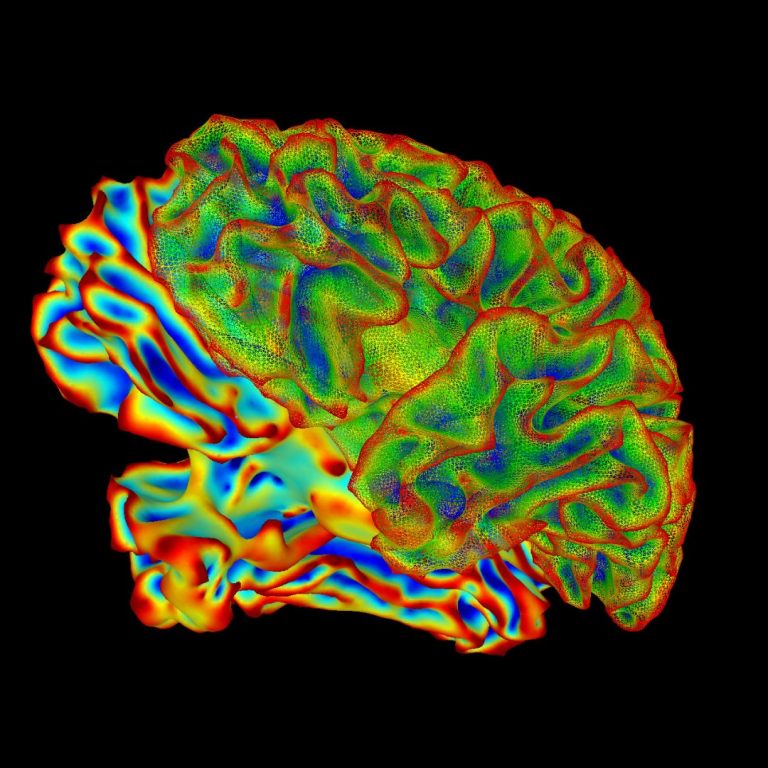
On Sept. 27, 2024, a study that evaluated the structural and functional connectivity correlates of fatigue in post-COVID…

On Sept. 20, 2024, study results released by Oregon Health & Science University (OHSU) researchers suggested placing defibrillation…

On Sept. 17, 2024, Boston Scientific announced the close of its acquisition of Silk Road Medical, a medical…

On Sep. 12, 2024, the the U.S. Food and Drug Administration (FDA) authorized the first over-the-counter (OTC) hearing…

On Sep. 9, 2024, Santo Fortunato, a professor at Luddy School of Informatics, reported that an international research…
On Aug. 23, 2024, A team of researchers from the University of Cologne announced they had created a…

On Aug. 14, 2024, a study co-led by Mass General Brigham found that one in four patients with…

On Aug. 2, 2024, the U.S. Food and Drug Administration approved Adaptimmune’s Tecelra (afamitresgene autoleucel), a gene therapy…

On Jul. 25, 2024, doctors from the Texas Heart Institute (THI), BiVACOR®, Baylor St. Luke’s Medical Center and…

In 1604, Dutch optician Zacharias Janssen is recognized as the inventor the compound microscope. Though rudimentary, the Janssen…

Montana Life Science Genealogy illustrates life science companies and non-profit research organization in the State of Montana by…
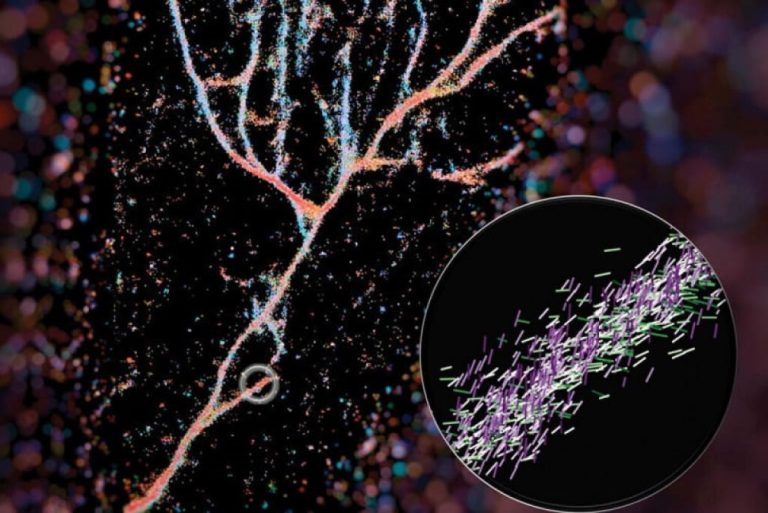
On Apr. 26, 2024, engineers at Washington University in St. Louis announced a new imaging technique that give…
On Apr. 24, 2024, surgeons at NYU Langone Health announced they had performed the first combined mechanical heart…

On Apr. 1, 2024, Otsuka Pharmaceutica and Click Therapeutics announced that the U.S. Food and Drug Administration (FDA)…

On Mar. 13, 2024, the National Science Board (NSB) published The State of U.S. Science and Engineering 2024,…

On Mar. 5, 2024, the U.S. Food and Drug Administration (FDA) cleared for marketing the first over-the-counter (OTC)…

On Mar. 4, 2024, Hugel America announced it received U.S. Food and Drug Administration (FDA) approval on its…
On Jan. 24, 2024, Oregon Health & Science University announced that Pamela Runner, 77, of Corvallis, became the…
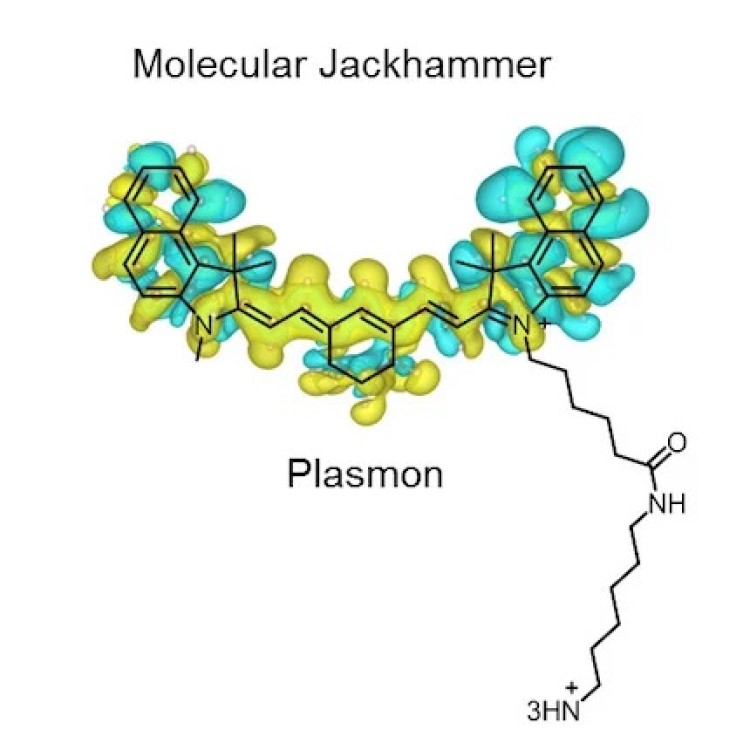
On Dec. 19, 2023, a research team led by Rice University reported they found that the atoms of…

On Nov. 17, 2023, Masimo announced that the Masimo W1™ medical watch has received FDA 510(k) clearance for over-the-counter…
On Oct. 27, 2023, the California Institute for Regenerative Medicine (CIRM) awarded $42.1 million to fund various translational…

On Sept. 26, 2023, the Advanced Research Projects Agency for Health (ARPA-H), an agency within the U.S. Department…

On Sept. 6, 2023, Oregon Health & Science University (OHSU) announced it had achieved a landmark in orthopaedic…

On Aug. 22, 2023, Alberta Innovates announced that more than $13.6 million will go to 19 partners, across…

On May 19, 2023, the U.S. Food and Drug Administration (FDA) cleared the Beta Bionics iLet ACE Pump…

On Mar. 14, 2023, National Institutes of Health (NIH) researchers announced they had developed a new imaging tool,…

On Mar. 6, 2023, Neuromod Devices announced that the U.S. Food and Drug Administration (FDA) had granted De…

On Feb. 14, 2023, the Pacific Economic Development Agency of Canada (PacifiCan), announced $14.5 million in funding for…
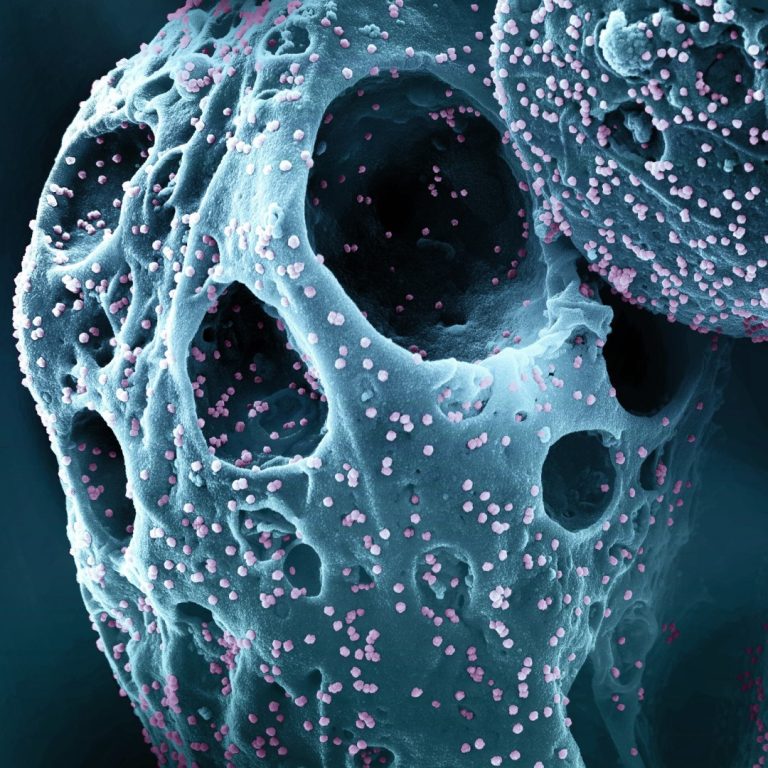
On Jan. 25, 2023, Roche and its subsidiary TIB Molbiol announced they had developed a COVID-19 PCR test…