The USDA confirmed COVID-19 in Hyenas at a Colorado Zoo
On Nov. 5, 2021, the USDA’s (USDA) National Veterinary Services Laboratories announced confirmation of SARS-CoV-2 in two spotted…

On Nov. 5, 2021, the USDA’s (USDA) National Veterinary Services Laboratories announced confirmation of SARS-CoV-2 in two spotted…

On Nov. 3, 2021, Novavax announced the company had filed for provisional approval of the vaccine to the…

On Nov. 1, 2021, Novavax announced the completion of its rolling submission to Health Canada for authorization of…

On Nov. 1, 2021, Novavax and Serum Institute of India announced that the National Agency of Drug and…

On Oct. 31, 2021, Moderna announced that the U.S. Food and Drug Administration (FDA) had notified the Company…
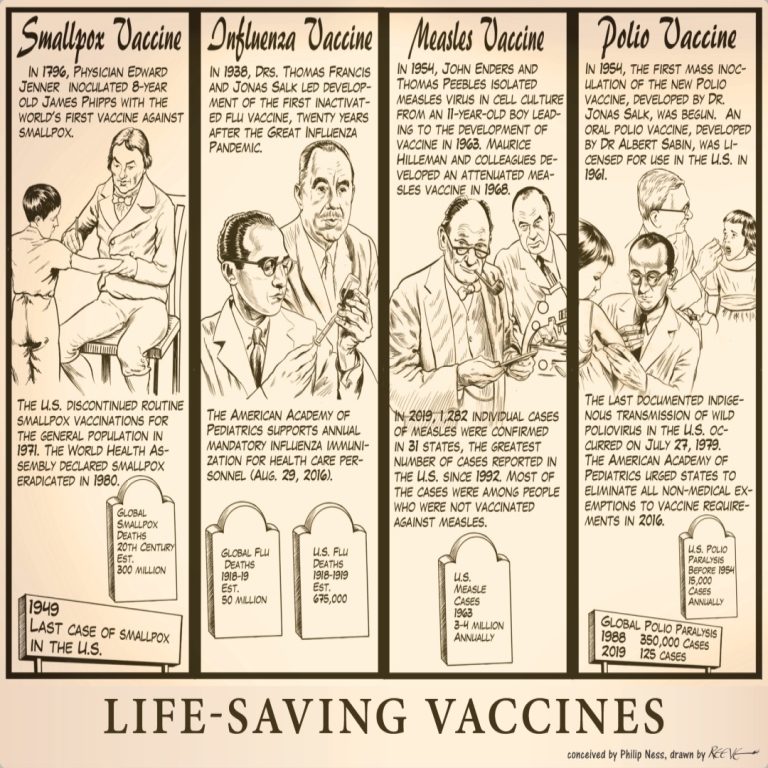
Our Life-Saving Vaccines cartoon illustrates four invaluable vaccines that have affected human civilization throughout history from smallpox and…

On Oct. 29, 2021, Novavax announced the completion of its rolling submission to the Therapeutic Goods Administration (TGA)…
On Oct. 27, 2021, Hologic announced that its Aptima SARS-CoV-2/Flu Assay was available for the simultaneous detection and…

On Oct. 27, 2021, Novavax announced the completion of its rolling regulatory submission to the U.K. Medicines and…

On Oct. 26, 2021, Moderna announced a new Memorandum of Understanding (MoU) to make up to 110 million…

On Oct. 26, 2021, Moderna announced that Swissmedic had authorized a booster dose of Spikevax, the Company’s vaccine…

On Oct. 25, 2021, Moderna announced positive interim data from the Phase 2/3 study, called the KidCOVE study,…

On Oct. 25, 2021, Moderna announced that the European Medicines Agency’s (EMA) Committee for Medicinal Products for Human…

On Oct. 22, 2021, the U.S. Centers for Disease Control and Prevention (CDC) reported two new U.S. human…

On Oct. 22, 2021, the U.S. Department of Veterans Affairs (VA) announced that it had begun offering Moderna…

On Oct. 20, 2021, Moderna announced that the U.S. Food and Drug Administration (FDA) had authorized for emergency…

On Oct. 20, 2021, Quest Diagnostics announced it had formed an agreement with the Texas Department of State…

On Oct. 14, 2021, Moderna confirmed that the U.S. Food and Drug Administration’s (FDA) Vaccines and Related Biological…

On Oct. 12, 2021, Moderna announced that Gavi, the Vaccine Alliance had exercised its option to purchase an…

On Oct. 12, 2021, Inovio Pharmaceuticals announced online preprint publication in MedRxiv of Phase 1 clinical data on…

On Oct. 11, 2021, Inovio Pharmaceuticals announced that it had received authorization from Colombia’s INVIMA, to conduct the…
On Oct. 7, 2021, PerkinElmer announced that the U.S. Food and Drug Administration (FDA) had issued Emergency Use…

On Oct. 7, 2021, Vaxart announced that a Duke University-led study published in bioRxiv showed that Vaxart’s (investigational…

On Oct. 7, 2021, Moderna announced it was building a state-of-the-art mRNA facility in Africa with the goal…

On Oct. 6, 2021, Washington University School of Medicine announced the start of a pediatric COVID-19 vaccine clinical…

On Oct. 5, 2021, Moderna announced that the European Medicines Agency (EMA) had authorized a third dose of…

On Oct. 4, 2021, Dynavax Technologies and the U.S. Department of Defense (DOD) announced Dynavax had executed an…
On Oct. 1, 2021, LabCorp announced that it had received Emergency Use Authorization (EUA) from the U.S. Food…

On Sept. 28, 2021, Sanofi announced that it had terminated plans for its own mRNA-based COVID-19 vaccine given…

On Sept. 27, 2021, Pfizer announced that the first participants have been dosed in a Phase 1 clinical…