Mylan to supply investigational antiviral remdesivir for potential treatment of COVID-19
On May 12, 2020, Mylan announced, as part of its ongoing efforts to support patients and public health…
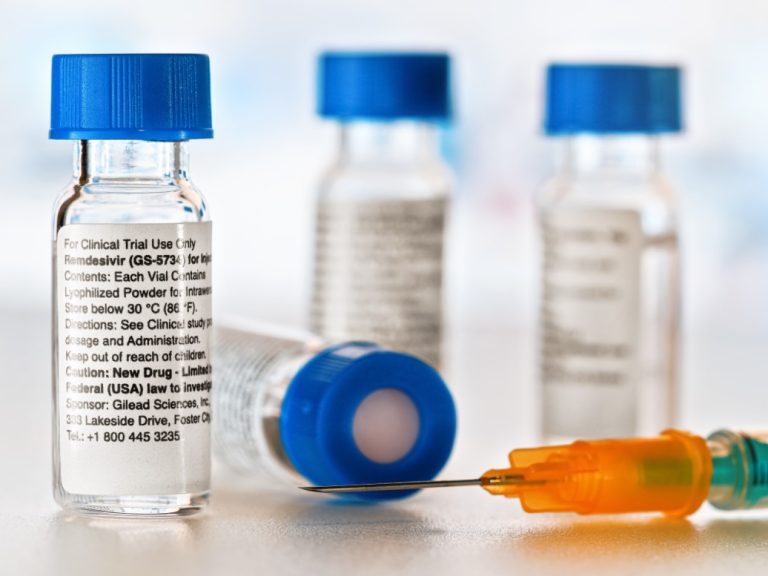
On May 12, 2020, Mylan announced, as part of its ongoing efforts to support patients and public health…

On May 11, 2020, the U.S. Food and Drug Administration (FDA) announced actions to accelerate the development of…

On May 11, 2020, a harmonized and collaborative approach to the clinical testing, scale-up and distribution of candidate…

On May 11, 2020, Abbott announced the U.S. Food and Drug Administration (FDA) issued an Emergency Use Authorization (EUA)…

On May 11, 2020, Todos Medical announced an exclusive distribution agreement with Gnomegen for the distribution of its…
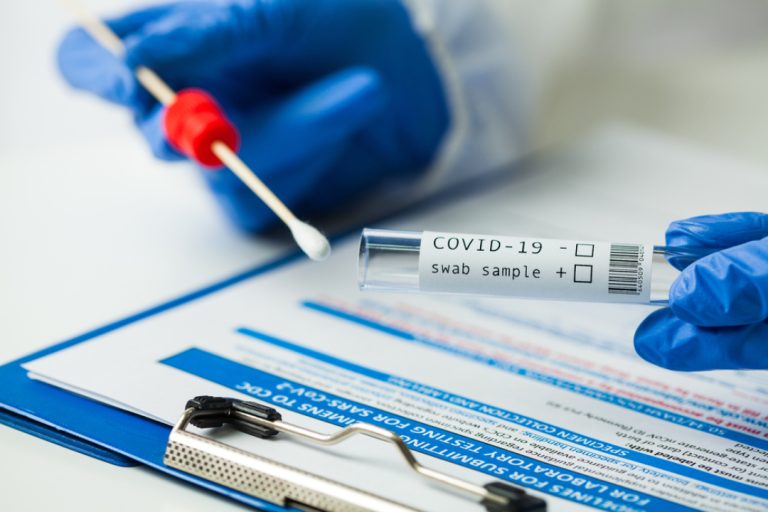
On May 9, 2020, the U.S. Food and Drug Administration (FDA) issued the first emergency use authorization (EUA)…

On May 8, 2020, the U.S. Food and Drug Administration (FDA) announced it had issued the first emergency…

On May 8, 2020, RedHill Biopharma announced the U.S. Food and Drug Administration (FDA) had approved its Investigational…

On May 8, 2020, Quidel announced it had received Emergency Use Authorization (EUA) from the FDA to market…

On May 7, 2020, OPTI Medical Systems, an IDEXX Laboratories subsidiary announced the U.S. Food and Drug Administration…

On May 6, 2020, PharmaCyte Biotech announced it had received Medical Devices Establishment Registration with the U.S. Food and…

On May 6, 2020, 3M and Ford announced that newly designed powered air-purifying respirators (PAPRs), developed are on…
On May 6, 2020, the U.S. Food and Drug Administration (FDA) approved Tabrecta (capmatinib) for the treatment of…

On May 5, 2020, ACON Laboratories announced the availability of its SARS-COV-2 IgG/IgM Rapid Test. ACON’s test is…

On May 4, 2020, bioMerieux announced that BioFire Diagnostics, its subsidiary specialized in syndromic infectious disease testing, has…

On May 4, 2020, Vir Biotech and Alnylam Pharma announced the selection of a development candidate for VIR-2703,…

On May 4, 2020, Cerus announced U.S. Food and Drug Administration (FDA) regulatory approval for manufacture of INTERCEPT…

On May 4, 2020, DNA Genotek, a subsidiary of OraSure Technologies, announced that its ORAcollect RNA kit (OR-100)…

On May 2, 2020, Roche announced that the U.S. Food and Drug Administration (FDA) has issued an Emergency…
On May 1, 2020, the Food and Drug Administration (FDA) approved daratumumab and hyaluronidase-fihj (DARZALEX FASPRO, Janssen Biotech.)…

On May 1, 2020, Gilead announced that the U.S. Food and Drug Administration (FDA) has granted emergency use…

On May 1, 2020, the U.S. Food and Drug Administration (FDA) issued an Emergency Use Authorization (EUA) for…

On Apr. 30, 2020, the FDA included, under the ventilator emergency use authorization (EUA), a ventilator developed by…

On Apr. 28, 2020, Chimerix announced it had received clearance from the FDA for a rolling submission of…

On Apr. 27, 2020, Mateon Therapeutics announced announced it has submitted an Investigational New Drug application to the…

On Apr. 27, 2020, Yield10 Bioscience announced that it has obtained a positive response from USDA-APHIS’s Biotechnology Regulatory Services…

On Apr. 27, 2020, Neurocrine Biosciences announced the U.S. Food and Drug Administration (FDA) had approved once-daily oral…

On Apr. 27, 2020, Moderna announced it had submitted an Investigational New Drug application to the U.S. Food…

On Apr. 27, 2020, Diffusion Pharmaceuticals announced the pre-IND submission to the FDA of a planned clinical program…

On Apr. 24, 2020, BioSig Technologies announced that subsidiary ViralClear had submitted an Investigational New Drug (IND) application…