Novavax filed in the EU for expanded conditional Marketing Authorization of COVID-19 vaccine as booster in individuals aged 18 and over
On May 20, 2022, Novavax announced the submission of a request to the European Medicines Agency (EMA) to…

On May 20, 2022, Novavax announced the submission of a request to the European Medicines Agency (EMA) to…

On May 18, 2022, Moderna and the nonprofit scientific research organization IAVI announced that the first participant screenings…

On May 18, 2022, the U.S. Department of Agriculture’s (USDA) Animal and Plant Health Inspection Service (APHIS) confirmed…
On May 18, 2022, LabCorp announced the receipt of Emergency Use Authorization (EUA) from the U.S. Food and…
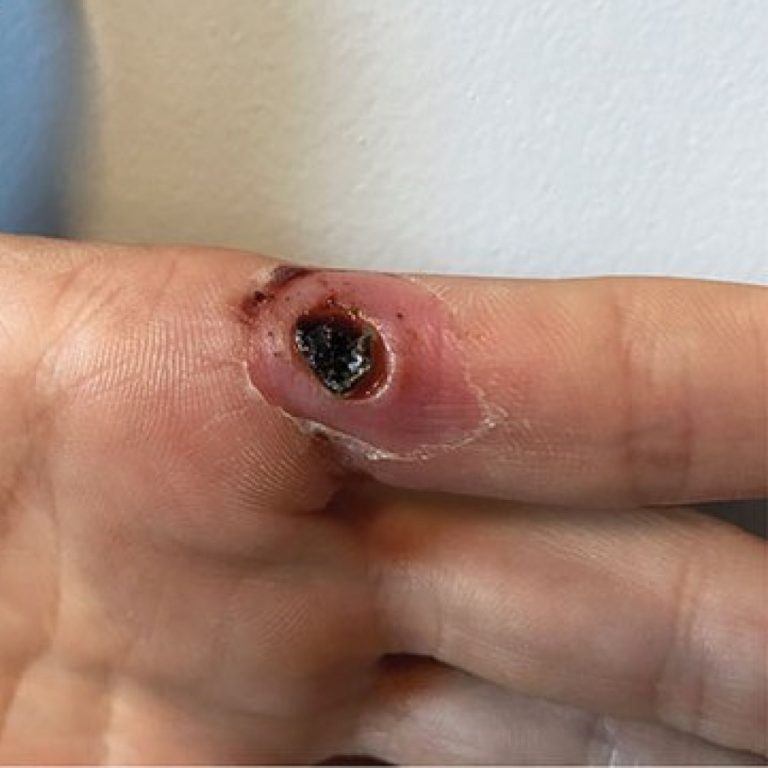
On May 18, 2022, scientists at the Centers for Disease Control and Prevention (CDC) announced that they were…

On May 13, 2022, the Tabula Sapiens Consortium announced it had published a molecular reference atlas for more…

On May 13, 2022, Novavax announced the submission of a request for emergency use authorization to Taiwan’s Food…

On May 11, 2022, Kaiser Permanente Washington Health Research Institute reported that new treatments for people at high…

On May 6, 2022, Novavax announced the submission of variations to the Australian Therapeutic Goods Agency (TGA) and…

On May 6, 2022, the U.S. Department of Agriculture’s Animal and Plant Health Inspection Service (APHIS) confirmed the…
On May 6, 2022, Novavax announced that deliveries of it’s COVID-19 vaccine continued around the world and the…

On May 2, 2022, Sorrento announced its Phase I study of intranasal STI-9199 (COVISHIELDTM) had been fully enrolled…

On May 1, 2022, the U.S. Department of Agriculture’s Animal and Plant Health Inspection Service confirmed the presence…

On Apr. 30, 2022, the U.S. Department of Agriculture’s (USDA) Animal and Plant Health Inspection Service (APHIS) confirmed…

On Apr. 29, 2022, Moderna announced its plan to build a state-of-the-art mRNA vaccine manufacturing facility in Quebec…

On Apr. 29, 2022, Moderna announced that it had submitted for a variation to the conditional marketing authorization…

On Apr. 29, 2022, the U.S. Department of Agriculture’s (USDA) Animal and Plant Health Inspection Service (APHIS) confirmed…

On Apr. 28, 2022, Moderna announced that it had submitted a request for emergency use authorization (EUA) for…

On Apr. 28, 2022, Centers for Disease Control and Prevention (CDC) reported that a person had tested positive…
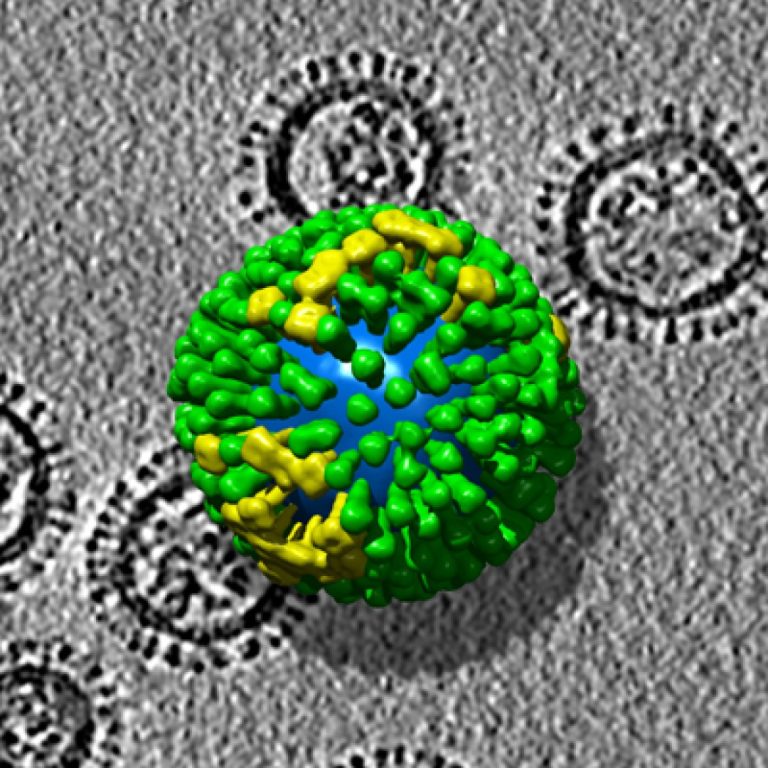
On Apr. 26, 2022, China’s National Health Commission reported a case of human infection with H3N8 avian influenza…

On Apr. 26, 2022, Pfizer and BioNTech submitted an application to the U.S. Food and Drug Administration (FDA)…

On Apr. 25, 2022, Gilead Sciences announced that the U.S. Food and Drug Administration (FDA) has approved a supplemental…

On Apr. 22, 2022, Stanford University announced it was awarded a $75 million gift from Nike co-founder Phil…

On Apr. 22, 2022, Novavax announced initial results from the Phase 1/2 clinical trial of its COVID-Influenza Combination…

On Apr. 22, 2022, the World Health Organization (WHO) made a strong recommendation for nirmatrelvir and ritonavir, sold…

On Apr. 21, 2022, Cocrystal Pharma announced a Non-Clinical Evaluation Agreement with the National Institute of Allergy and…

On Apr. 20, 2022, Novavax announced initial results from the Phase 1/2 clinical trial of its COVID-Influenza Combination…

On Apr. 20, 2022, Tonix Pharmaceuticals announced the results of a retrospective observational database study in over 50,000…

On Apr. 19, 2022, Novavax announced that its partner, Takeda, received manufacturing and marketing approval from the Japan…

On Apr. 19, 2022, Moderna announced new clinical data on its bivalent COVID-19 booster platform including data on…