iBio Discovers Novel Antibody Targeting Activin E in Collaboration with AstralBio
On Jan. 7, 2025, IBIO an AI-driven innovator of precision antibody immunotherapies, in collaboration with AstralBio announced the…

On Jan. 7, 2025, IBIO an AI-driven innovator of precision antibody immunotherapies, in collaboration with AstralBio announced the…

On Jan. 7, 2025, researchers at the Francis Crick Institute, MRC Harwell Institute and UCL Queen Square Institute…
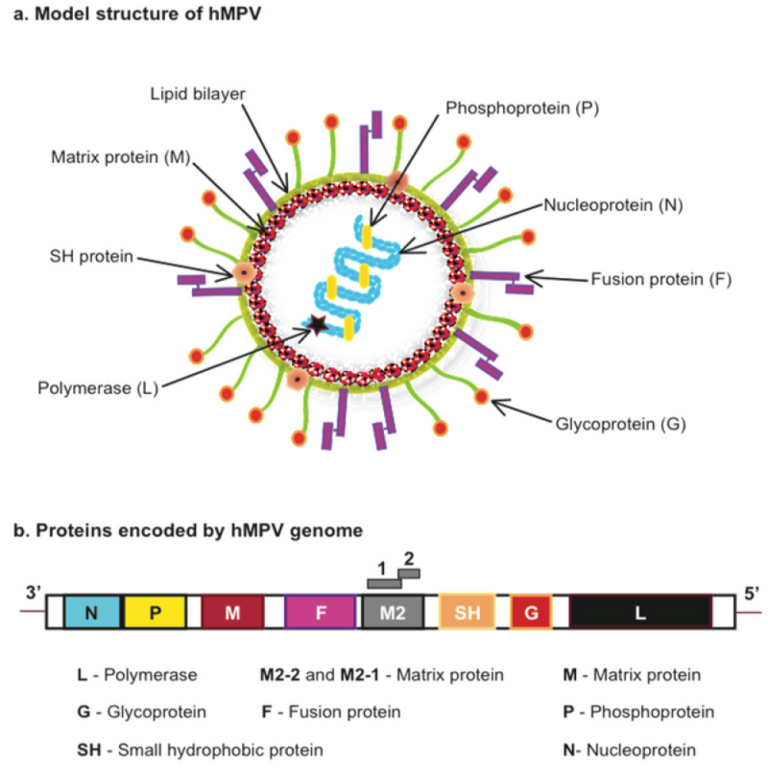
On Jan. 7, 2025, the World Health Organization (WHO) reported that trends in acute respiratory infections (ARI) in…
On Jan. 7, 2025, scientists at The University of Queensland (UQ) and the University of Sydney announced they…

On Jan. 7, 2025, researchers at Tufts University and Oxford University announced they have uncovered mechanisms connecting trauma…
On Jan. 6, 2025, the Louisiana Department of Health reports the patient who had been hospitalized with the…

On Jan. 6, 2025, the U.S. Food and Drug Administration (FDA) issued guidance for industry on the action…

On Jan. 3, 2025, the California Department of Public Health (CDPH) reported that Whooping cough (pertussis) activity in…
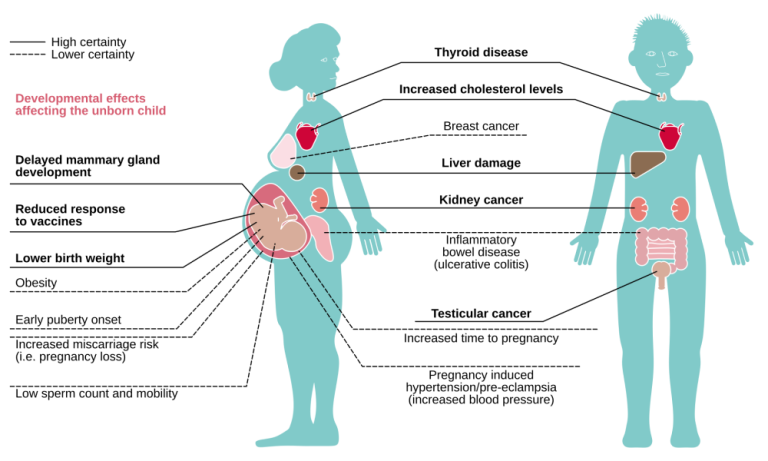
On Jan. 3, 2025, the U.S. Environmental Protection Agency announced the automatic addition of nine per- and polyfluoroalkyl…

On Jan. 3, 2025, the Centers for Disease Control and Prevention (CDC) reported that during Christmas week, respiratory…

On Jan. 3, 2025, the U.S. Department of Health and Human Services (HHS) announced it would award $306…

On Jan. 3, 2025, the U.S. General Dr. Vivek Murthy released a new Surgeon General’s Advisory on Alcohol…

On Jan. 2, 2025, SIGA Technologies announced that its antiviral treatment TEPOXX (tecovirimat 200 mg capsules), marketed as…
On Jan. 2, 2025, the U.S. Centers for Disease Control and Prevention (CDC) reported an increased incidence of…

On Jan. 2, 2025, an international study led by researchers at Karolinska Institutet in Sweden shows that AI-based…

On Jan. 1, 2025, a study led by the Francis Crick Institute showed that waves of human migration…
On Dec. 31, 2024, the U.S. Centers for Disease Control and Prevention released a study of the largest…
On Dec. 31, 2024, a study was published of patients with kidney disease who are at high risk…
On Dec. 30, 2024, the Oregon Health Authority reported it had registered 1,105 cases of pertussis—also known as…

On Dec. 30, 2024, the Maricopa County Department of Public Health (MCDPH), as part of routine wastewater surveillance…

On Dec. 30, 2024, a study published in JAMA Network Open found that the estimated effectiveness of at least…

On Dec. 30, 2024, the Government of China announced it had approved five gene-edited crop varieties and 12…
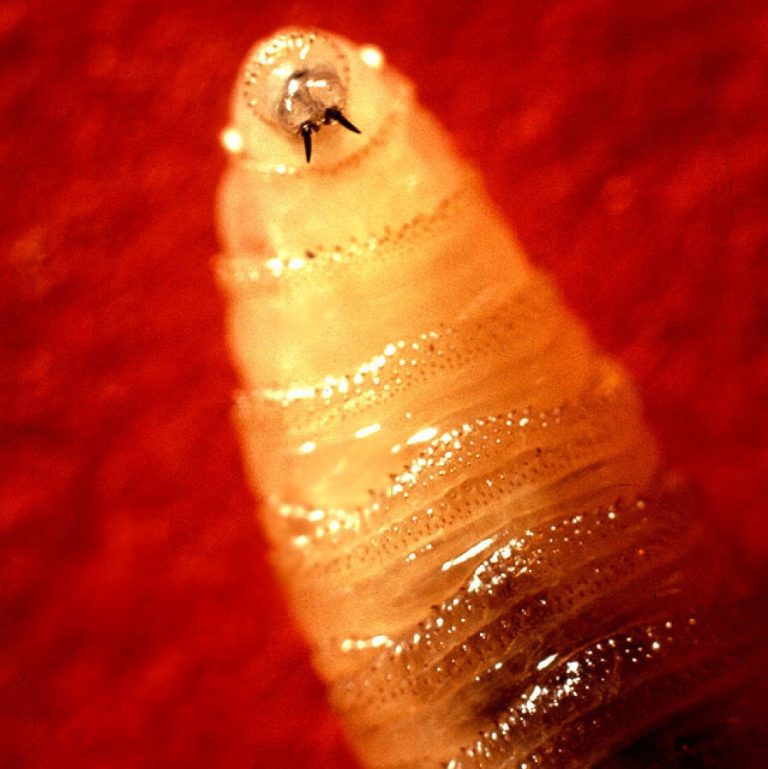
On Dec. 30, 2024, Texas Parks and Wildlife Department (TPWD) asked hunters and other outdoor enthusiasts in South…
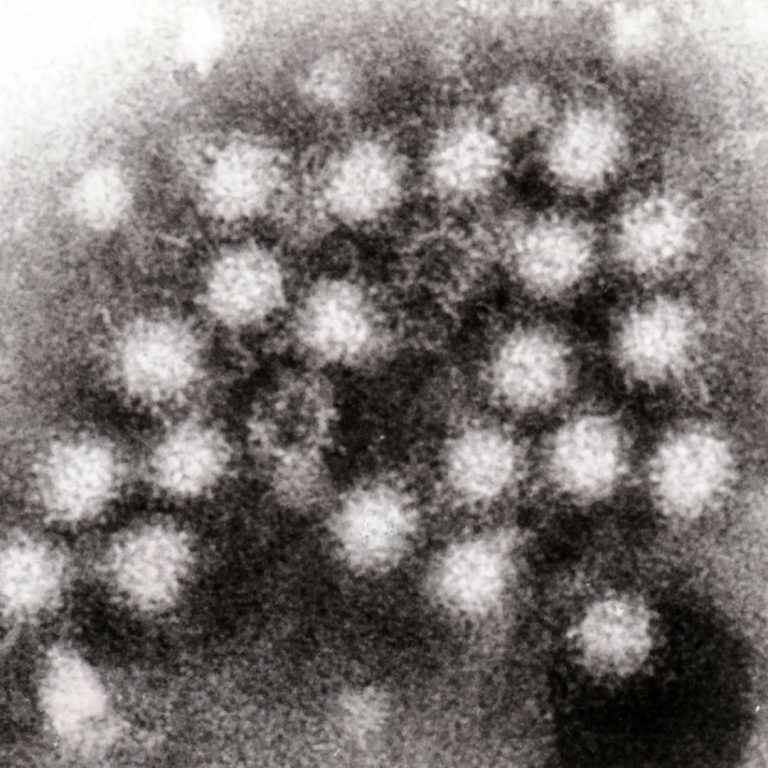
On Dec. 28, 2024, the U.S. Centers for Disease Control and Prevention (CDC) reported that cases of a…

On Dec. 27, 2024, BioNTech announced it had entered into two separate settlement agreements with the U.S. National…

On Dec. 26, 2024, the Oregon Department of Agriculture (ODA) announced it is alerting pet owners that samples…

On Dec. 24, 2024, the World Health Organization (WHO) announced that SARS-CoV-2, the virus that causes COVID-19, largely…

On Dec. 24, 2024, the Minnesota Department of Natural Resources (MNDNR) is investigating a number of wild waterfowl…
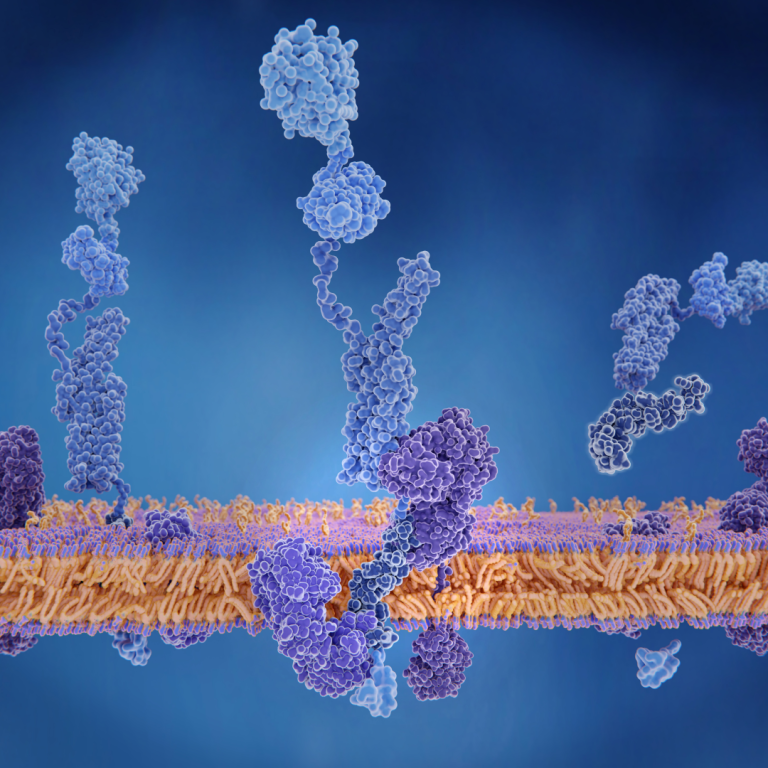
On Dec. 23, 2024, researchers with the Advanced Science Research Center at the CUNY Graduate Center (CUNY ASRC)…

On Dec. 23, 2024, an Annenberg Public Policy Center (APPC) health survey conducted in November 2024 showed that…