Research collaboration launched to focus on repurposing existing anti-viral therapies to treat COVID-19
On Feb. 4, 2022, a new ᆪ1.6m collaborative project was launched to rapidly identify new treatments for COVID-19….

On Feb. 4, 2022, a new ᆪ1.6m collaborative project was launched to rapidly identify new treatments for COVID-19….

On Feb. 4, 2022, pet hamsters probably carried the Delta variant of SARS-CoV-2 into Hong Kong and sparked…

On Feb. 4, 2022, the Advisory Committee on Immunization Practices (ACIP) issued a standard recommendation for use of…

On Feb. 3, 2022, Novavax announced that the Medicines and Healthcare products Regulatory Agency had granted conditional marketing…

On Feb. 3, 2022, Novavax announced that New Zealand’s Medsafe had been granted provisional approval of NVX-CoV2373, Novavax’…

On Feb. 3, 2022, T2 Biosystems announced that its T2SARS-CoV-2 Panel detected Omicron COVID-19 subvariants BA.1, BA.2, and…
On Feb. 2, 2022, NIAID scientists announced the new Pandemic Preparedness Plan aimed to support critical basic and…

On Feb. 2, 2022, the U.S. Food and Drug Administration announced it had approved the first generic of…

On Feb. 2, 2022, a new analysis of the first two patients treated in a clinical trial with…
On Feb. 2, 2022, President Biden announced a reignition of the Cancer Moonshot, that highlighted new goals: to…

On Feb. 1, 2022, Pfizer and BioNTech announced that following a request from the U.S. Food and Drug…

On Jan. 31, 2022, Novavax announced that it had submitted a request to the U.S Food and Drug…
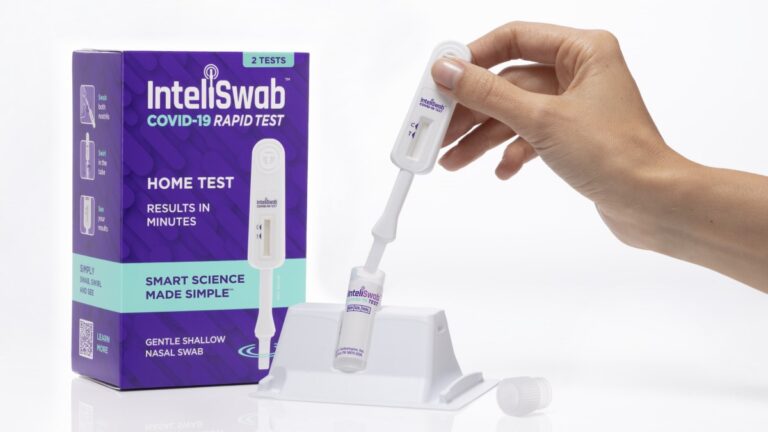
On Jan. 31, 2022, OraSure Technologies announced that its InteliSwab COVID-19 rapid tests had been authorized by the…

On Jan. 31, 2022, Moderna announced the U.S. Food and Drug Administration (FDA) had approved the Biologics License…

On Jan. 31, 2022, Veru announced that the U.S. Food and Drug Administration (FDA) had granted Fast Track…

On Jan. 28, 2022, Merck and Ridgeback Biotherapeutics announced data from six preclinical studies demonstrating that molnupiravir, an…

On Jan. 28, 2022, Novavax and Israel’s Ministry of Health announced an agreement for the purchase of NVX-CoV2373,…

On Jan. 28, 2022, the WHO announced that COVID-19 information had reached 1,292,209 people through Viamoメs 3-2-1 Platform…

On Jan. 27, 2022, Cocrystal Pharma announced that it had selected two investigational novel antiviral drug candidates for further…
On Jan. 27, 2022, NIAID announced that a clinical trial found that the combination of remdesivir plus a…

On Jan. 26, 2022, scientists at Tufts and Harvard University’s Wyss Institute reported they were able to trigger…

On Jan. 26, 2022, a former University of British Columbia post-doctoral research fellow led an international research team…

On Jan. 26, 2022, the NIH announced that adults who had previously received a full regimen of any…

On Jan. 26, 2022, Moderna announced that the first participant had been dosed in the Phase 2 study…

On Jan. 25. 2022, a report published in PNAS provided strong evidence of extensive SARS-CoV-2 infection of white-tailed…

On Jan. 25, 2022, researchers at the University of Missouri announced they had identified the highly prevalent, specific…

On Jan. 25, 2022, Cepheid announced that Health Canada had issued Cepheid a medical device license for Xpert…

On Jan. 25, 2022, Pfizer and BioNTech announced the initiation of a clinical study to evaluate the safety,…

On Jan. 25, 2022, the U.S. Department of Agricultureメs National Veterinary Services Laboratories announced a study with zoos…

The Evolution of CRISPR illustrates its climb out of the primordial ocean on the backs of eukaryotic cells…