Tevogen Bio submitted IND for T cell treatment in COVID-19
On Oct. 14, 2020, Tevogen Bio announced that its Investigational New Drug (IND) application to develop a COVID-19…

On Oct. 14, 2020, Tevogen Bio announced that its Investigational New Drug (IND) application to develop a COVID-19…
On Oct. 14, 2020, Regeneron announced that the FDA had approved Inmazeb (atoltivimab, maftivimab and odesivimab-ebgn) for the…
On Oct. 14, 2020, 3M and Discovery Education announced they had named 14-year-old Anika Chebrolu from Frisco, Texas,…

On Oct. 14, 2020, Sorrento Therapeutics announced receipt of clearance from the Brazilian regulatory agency (ANVISA) to proceed…
On Oct. 14, 2020, the World Health Organization (WHO) announced that prior to the COVID-19 pandemic, many countries…
On Oct. 14, 2020, recognizing the urgent need for new tools to combat vector-borne diseases (VBDs), and in…
On Oct. 14, 2020, the FDA approved Regeneron Pharmaceutical’s Inmazeb (atoltivimab, maftivimab, and odesivimab-ebgn), a mixture of three…

On Oct. 14, 2020, Vaxart announced that the FDA had completed its review of the Companyメs Investigational New…

On Oct. 14, 2020, the U.S. Department of Health and Human Services (HHS) and Department of Defense (DOD)…

On Oct. 14, 2020, Thermo Fisher Scientific announced plans to develop two new sterile filling lines in Singapore…

On Oct. 14, 2020, Moderna announced that it had received written confirmation from the European Medicines Agency (EMA)…

On Oct. 13, 2020, Howard Hughes Medical Institute Investigator Leslie Vosshall’s team at The Rockefeller University and colleagues…

On Oct. 13, 2020, the Department of Defense (DOD), on behalf of the U.S. Department of Health and…

On Oct. 13, 2020, Medigen Vaccine Biologics (MVC) and Dynavax Technologies announced that MVC had obtained a Taiwan…
On Oct. 13, 2020, Humanigen announced that the National Institute of Allergy and Infectious Diseases (NIAID), had launched…

On Oct. 13, 2020, the National Institute on Drug Abuse (NIDA) and nine other institutes, all part of…

On Oct. 13, 2020, Roche announced that it intended to launch a high-volume SARS-CoV-2 Antigen test as an…
On Oct. 13, 2020, NeoGenomics announced plans to open a state-of-the-art research laboratory in China in association with…

On Oct. 13, 2020, the National Institute of Allergy and Infectious Diseases (NIAID), launched a study designed to…

On Oct. 13, 2020, RedHill Biopharma announced collaborations with two specialist pharmaceutical manufacturers in Europe and Canada to…

On Oct. 13, 2020, Apogenix announced that the first patient has been enrolled in the ASUNCTIS trial. The…

On Oct. 13, 2020, Cue Health announced a $481 Million award from the Department of Defense (DOD), on…

On Oct. 13, 2020, Eli Lilly announced that the government-sponsored clinical trial of its COVID-19 antibody treatment similar…

On Oct. 13, 2020, Roswell Park Comprehensive Cancer Center announced that it had been approved to administer the…

On Oct. 13, 2020, Moderna announced initiation of a rolling submission to Health Canada for mRNA-1273, the Company’s…

On Oct. 12, 2020, the Washington University School of Medicine in St. Louis announced a $14.6 million grant…
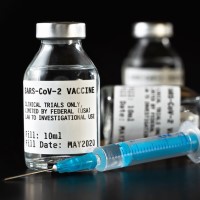
On Oct. 12, 2020, Johnson & Johnson announced temporarily paused further dosing in all our COVID-19 vaccine candidate…

On Oct. 12, 2020, Thermo Fisher Scientific introduced two new SARS-CoV-2 antibody tests: the Thermo Scientific OmniPATH COVID-19…

On Oct. 9, 2020, OPKO Health subsidiary BioReference Labs, New York City, the Department of Health, New York…

On Oct. 9, 2020, Pfizer and BioNTech announced the initiation of a rolling submission to Health Canada for…