Innovation Pharmaceuticals Brilacidin tested as drug and vaccine at different institutions
On Mar. 17, 2020, Innovation Pharma announced details on the Material Transfer Agreement signed with a leading public…

On Mar. 17, 2020, Innovation Pharma announced details on the Material Transfer Agreement signed with a leading public…

On Mar. 17, 2020, BioReference Laboratories, an OPKO Health company, announced a collaboration with the New York City…
On Mar. 17, 2020, Pfizer and BioNTech announced the companies have agreed to a letter of intent regarding…

On Mar. 17, 2020, Regeneron announced that its scientists had isolated hundreds of virus-neutralizing, fully human antibodies from…
On Mar. 17, 2020, Todos Medical announced a non-exclusive distribution agreement with 3D Biomedicine Science & Technology, a…
On Mar. 17, 2020, MannKind announced it was adjusting research and development resources that were reserved for its…
On Mar. 16, 2020, the Hormel Institute, University of Minnesota announced published research concerning remnants of ancient viruses…
On Mar. 16, 2020, the National Institutes of Health (NIH announced that a Phase 1 clinical trial evaluating…
On Mar. 16, 2020, new insights into the architecture of the brain were revealed by scientists at the…

On Mar. 16, 2020, Aimmune Therapeutics announced that the first patients in the U.S. were being treated with…

On Mar. 16, 2020, Moderna announced that the first participant had been dosed in the Phase 1 study…

On Mar. 16, 2020, Hologic announced the U.S. Food and Drug Administration (FDA) had granted Emergency Use Authorization…

On Mar. 16, 2020, BD (Becton, Dickinson and Company) announced the companies had submitted Emergency Use Authorization requests…

On Mar. 16, 2020, Sanofi and Regeneron Pharma announced they have started a clinical program evaluating Kevzara (sarilumab)…

On Mar. 16, 2020, Meridian Bioscience announced its increased commitment to the global fight against COVID-19 by making…

On Mar. 15, 2020, Iceland health authorities and deCode Genetics announced they began comprehensive screening for the virus…
On Mar. 15, 2020, the National Institutes of Health (NIH) informed its staff that it had its first…
On Mar. 15, 2020, Fosun Pharma and BioNTech announced a strategic development and commercialization collaboration to advance BioNTech’s…

On Mar. 13, 2020, Roche announced the U.S. Food and Drug Administration (FDA) has issued an Emergency Use Authorization…

On Mar. 13, 2020, Thermo Fisher Scientific announced the U.S. Food and Drug Administration (FDA) has issued an emergency…

On Mar. 13, 2020, declared a COVID-related emergency under Section 501(b) of the Stafford Act [18]. Under the…

On Mar. 13, 2020, Washington University in St. Louis announced the first long-term study of Bacillus thuringiensis (Bt)…
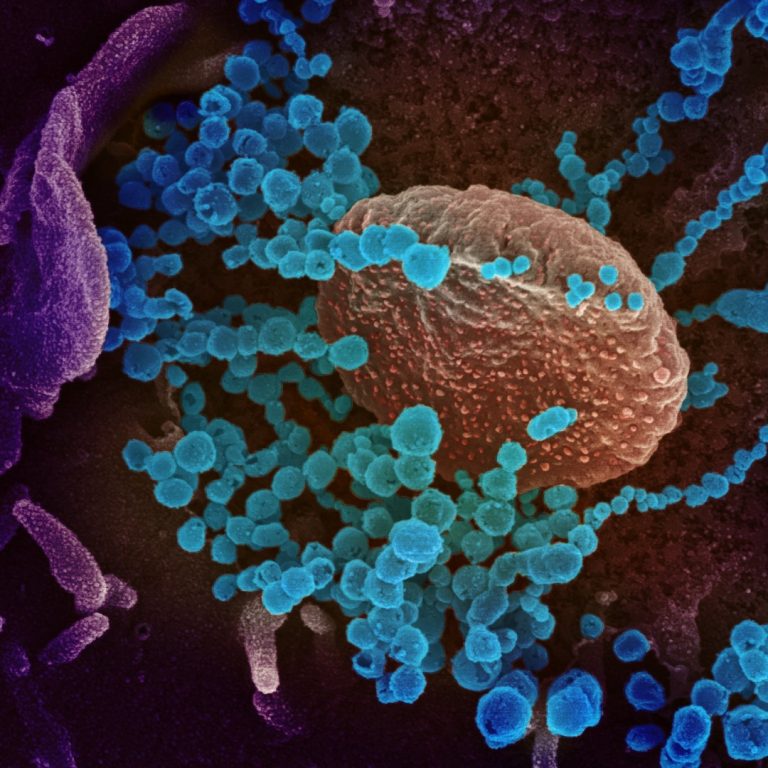
On Mar. 13, 2020, Enanta Pharmaceuticals announced it had initiated a program to discover direct-acting antiviral drug candidates…

On Mar. 13, 2020, IDEXX Laboratories announced the company had seen no positive results in pets to date…

On Mar. 13, 2020, Johnson & Johnson announced that its Janssen Pharmaceutical Companies have entered a collaboration with…

On Mar. 13, 2020, BioReference Laboratories, an OPKO company, announced it had begun accepting specimens for testing of…

On Mar. 13, 2020, Luminex announced that four independent clinical laboratories had validated laboratory developed tests (LDTs) for…

On Mar. 13, 2020, PATH and the Centers for Disease Control and Prevention (CDC) announced a collaboration with…

On Mar. 13, 2020, researchers at the National Eye Institute (NEI) reported finding a protein that normally deposits…
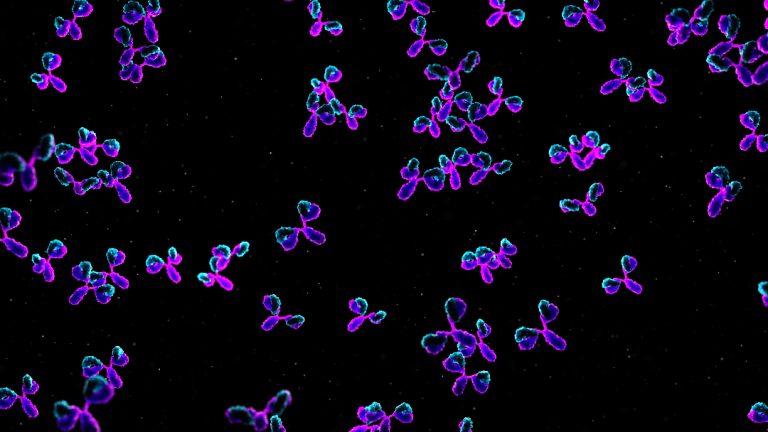
On Mar. 12, 2020, Vir Biotech announced it had signed a letter of intent with Biogen for the…