OHSU received more than $11 million to continue brain, cognitive development research
On Apr. 17, 2020, the National Institutes of Health (NIH) announced nearly $290 million of new funding for…
On Apr. 17, 2020, the National Institutes of Health (NIH) announced nearly $290 million of new funding for…
On Apr. 17, 2020, the University of Washington Medicine announced that a new clinical blood test will be…

On Apr. 17, 2020, as part of Project Orbis, the U.S. Food and Drug Administration (FDA) approved Seagen’s…
On Apr. 17, 2020, five projects from across Oxford University’s Medical Sciences Division were among twenty-one new studies…

On Apr. 17, 2020, Roche announced the development and upcoming launch of its Elecsys Anti-SARS-CoV-2 serology test to…

On Apr. 17, 2020, Kaiser Permanente Washington Health Research Institute (KPWHRI) announced that older adults were being enrolled…

On Apr. 17, 2020, Sinovac Biotech announced that the Company had commenced Phase I clinical trial, a randomized,…
On Apr. 17, 2020, after promising COVID-19 treatments in Germany, France, Italy, Spain and the U.S,, ExThera Medical’s…

On Apr. 17, 2020, Incyte announced the initiation of RUXCOVID, a global, randomized, double-blind, placebo-controlled phase 3 clinical…
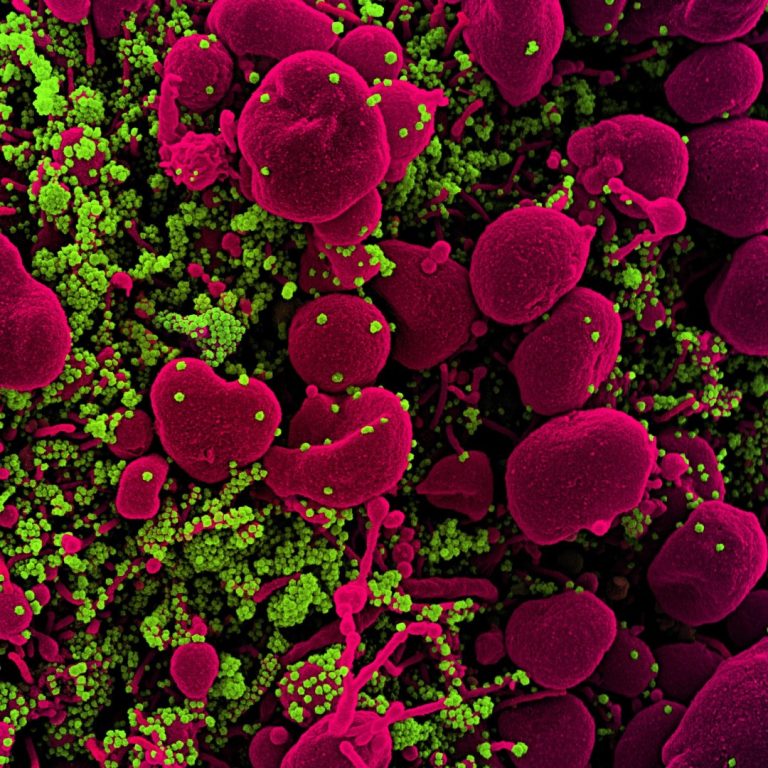
On Apr. 17, 2020, researchers at Montefiore Health System and Albert Einstein College of Medicine may have solved…

On Apr. 17, 2020, Emory University announced it had begun enrolling adults over age 55 in the clinical…

On Apr. 17, 2020, Celdara Medical announced that it had entered into a Cooperative Research and Development Agreement…
On Apr. 16, 2020, Moderna announced an agreement for a commitment of up to $483 million from the…
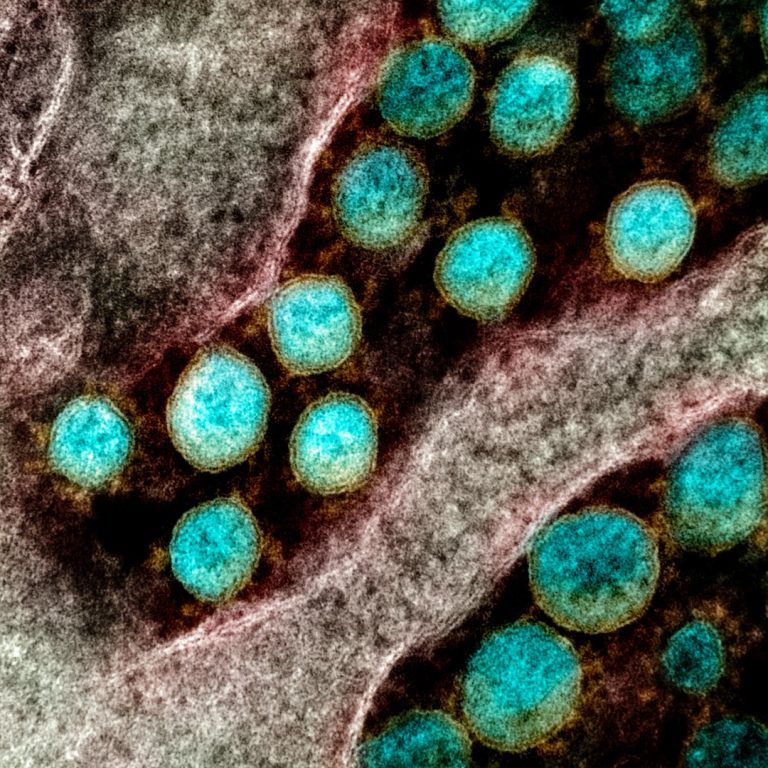
On Apr. 16, 2020, a team of researchers at the McGovern Institute for Brain Research at MIT, the…

On Apr. 16, 2020, CHF Solutions announced increased utilization of its Aquadex therapy in patients affected by COVID-19….

On Apr. 16, 2020, Soligenix announced it has executed an agreement for the exclusive worldwide license of CoVaccine…

On Apr. 16, 2020, Regeneron announced the FDA has accepted for Priority Review a new Biologics License Application…

On Apr. 16, 2020, OpGen announced that a study on the feasibility and potential of antibiotic susceptibility testing…
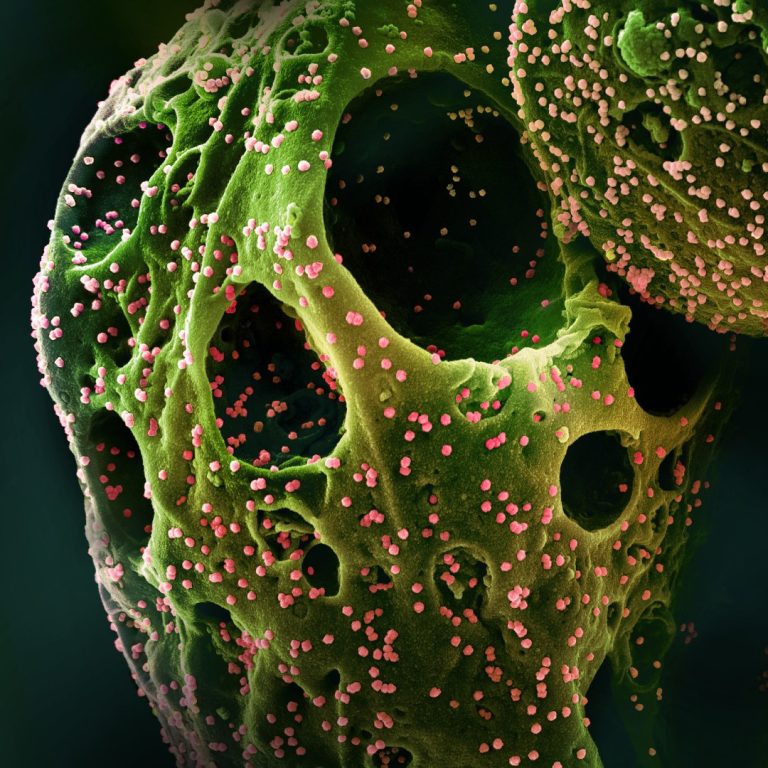
On Apr. 16, 2020, Atossa Therapeutics announced a new drug development program called COVID-19 HOPE. The program used…

On Apr. 16, 2020, the International Vaccine Institute (IVI) announced that the Coalition for Epidemic Preparedness Innovations (CEPI)…

On Apr. 16, 2020, Mammoth Biosciences announced the publication of a study demonstrating the power of its platform…

On Apr. 16, 2020, Dynavax and Sinovac Biotech, a leading provider of biopharmaceutical products in China, announced a…

On Apr. 16, 2020, Scripps Research announced that researchers at Calibr, Scripp’s drug development division, had established an…

On Apr. 16, 2020, BioSig Technologies announced that its subsidiary ViralClear Pharmaceuticals, had updated its clinical development program…

On Apr. 16, 2020, Biogen, Broad Institute of MIT and Harvard, and Partners HealthCare announced a consortium that…

On Apr. 16, 2020, Pulmatrix announced a collaboration and license agreement with Sensory Cloud. Under the terms of…

On Apr. 15, 2020, the FDA announced it granted Emergency Use Authorization to Ortho Clinical Diagnosticsメ total antibody…

On Apr. 15, 2020, Todos Medical announced the FDA has accepted Todos Medical’s application for Medical Device Establishment…

On Apr. 15, 2020, Vanda Pharma and Northwell Health’s research arm, The Feinstein Institutes for Medical Research, announced…

On Apr. 15, 2020, Bellicum Pharma announced closing of the sale of its Houston facility to The University…