NIAID Pandemic Preparedness Plan targeted ‘prototype’ and priority pathogens
On Feb. 2, 2022, NIAID scientists announced the new Pandemic Preparedness Plan aimed to support critical basic and…
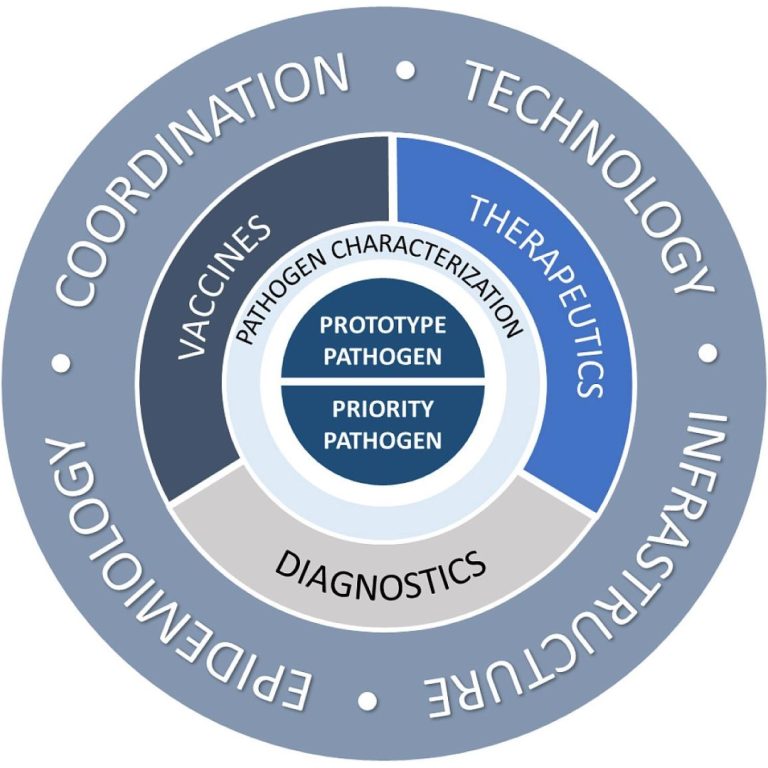
On Feb. 2, 2022, NIAID scientists announced the new Pandemic Preparedness Plan aimed to support critical basic and…

On Feb. 2, 2022, the U.S. Food and Drug Administration announced it had approved the first generic of…

On Feb. 2, 2022, a new analysis of the first two patients treated in a clinical trial with…
On Feb. 2, 2022, President Biden announced a reignition of the Cancer Moonshot, that highlighted new goals: to…

On Feb. 1, 2022, Pfizer and BioNTech announced that following a request from the U.S. Food and Drug…

On Jan. 31, 2022, Veru announced that the U.S. Food and Drug Administration (FDA) had granted Fast Track…
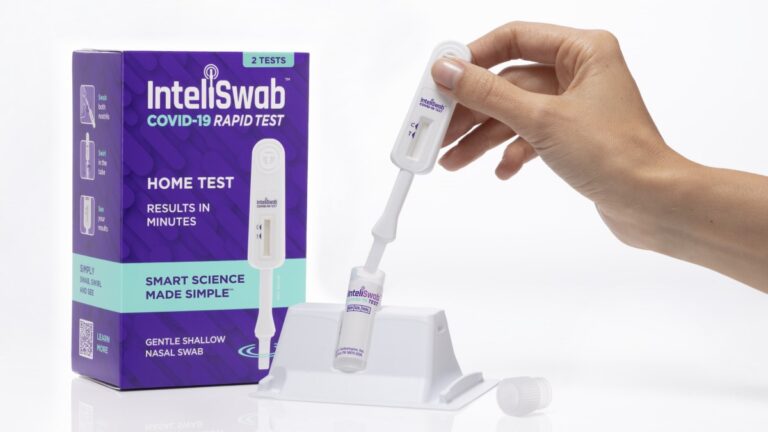
On Jan. 31, 2022, OraSure Technologies announced that its InteliSwab COVID-19 rapid tests had been authorized by the…

On Jan. 31, 2022, Novavax announced that it had submitted a request to the U.S Food and Drug…

On Jan. 31, 2022, Moderna announced the U.S. Food and Drug Administration (FDA) had approved the Biologics License…

On Jan. 28, 2022, Novavax and Israel’s Ministry of Health announced an agreement for the purchase of NVX-CoV2373,…

On Jan. 28, 2022, the WHO announced that COVID-19 information had reached 1,292,209 people through Viamoメs 3-2-1 Platform…

On Jan. 28, 2022, Merck and Ridgeback Biotherapeutics announced data from six preclinical studies demonstrating that molnupiravir, an…
On Jan. 27, 2022, NIAID announced that a clinical trial found that the combination of remdesivir plus a…

On Jan. 27, 2022, Cocrystal Pharma announced that it had selected two investigational novel antiviral drug candidates for further…

On Jan. 26, 2022, Moderna announced that the first participant had been dosed in the Phase 2 study…

On Jan. 26, 2022, the NIH announced that adults who had previously received a full regimen of any…

On Jan. 26, 2022, scientists at Tufts and Harvard University’s Wyss Institute reported they were able to trigger…

On Jan. 26, 2022, a former University of British Columbia post-doctoral research fellow led an international research team…

On Jan. 25, 2022, Cepheid announced that Health Canada had issued Cepheid a medical device license for Xpert…

On Jan. 25, 2022, researchers at the University of Missouri announced they had identified the highly prevalent, specific…

On Jan. 25, 2022, the U.S. Department of Agricultureメs National Veterinary Services Laboratories announced a study with zoos…

On Jan. 25, 2022, Pfizer and BioNTech announced the initiation of a clinical study to evaluate the safety,…

On Jan. 25. 2022, a report published in PNAS provided strong evidence of extensive SARS-CoV-2 infection of white-tailed…

The Evolution of CRISPR illustrates its climb out of the primordial ocean on the backs of eukaryotic cells…

On Jan. 24, 2022, the University of Oxford announced that a third booster dose of either the ChAdOx1…

On Jan. 24, 2022, Atara Biotherapeutics, a leader in T-cell immunotherapy, announced that it had leveraged its novel…

On Jan. 24, 2022, Anixa Biosciences announced that the company and its partner, MolGenie, had synthesized a compound…
On Jan. 24, 2022, Pfizer and BioNTech announced the publication of new results from two laboratory studies demonstrating…

On Jan. 20, 2022, Gilead Sciences announced that the U.S. Food and Drug Administration had granted expedited approval…

On Jan. 20, 2022, a clinical trial funded by the National Institutes of Health (NIH) found that giving…