BD announced second FDA Emergency Use Authorization, CE Mark for new COVID-19 molecular diagnostic for global use
On Apr. 13, 2020, BD (Becton, Dickinson) announced the U.S. Food and Drug Administration (FDA) had granted Emergency…

On Apr. 13, 2020, BD (Becton, Dickinson) announced the U.S. Food and Drug Administration (FDA) had granted Emergency…

On Apr. 13, 2020, Athersys announced the FDA has authorized the Company to initiate a Phase 2/3 pivotal…

On Apr. 13, 2020, Aegis Sciences announced serving as a regional reference laboratory for COVID-19 testing being directed…

On Apr. 10, 2020, a new study has begun recruiting at the NIH in Bethesda, Maryland, to determine…

On Apr. 10, 2020, STERIS announced that the FDA issued an Emergency Use Authorization (EUA) for respirator decontamination….

On Apr. 10, 2020, the U.S. Department of Veterans Affairs (VA) established the VA History Office (VAHO) to…

On Apr. 10, 2020, BenevolentAI announced that baricitinib, which it had identified as a potential treatment for COVID-19,…
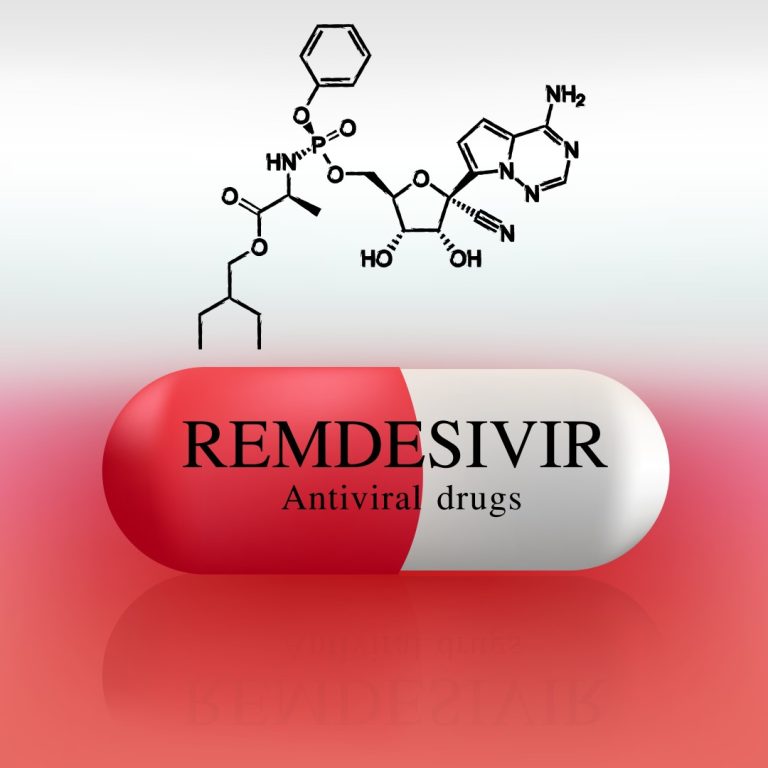
On Apr. 10, 2020, Gilead Sciences announced results from a cohort analysis of 53 patients hospitalized with severe…

On Apr. 10, 2020, Terumo BCT and Marker Therapeutics announced the FDA issued an Emergency Use Authorization (EUA)…

On Apr. 10, 2020, Eli Lilly announced it had entered into an agreement with the National Institute of…
On Apr. 10, 2020, Oregon Health & Science University (OHSU) announced that a 68-year-old Oregonian was recovering at…

On Apr. 10, 2020, the U.S. Food and Drug Administration (FDA) approved selumetinib (KOSELUGO, AstraZeneca) for pediatric patients, 2…
On Apr. 9, 2020, the first participants were enrolled in the trial at Vanderbilt University Medical Center, Nashville,…

On Apr. 9, 2020, FUJIFILM announced initiation of a U.S. phase II clinical trial to evaluate the safety…
On Apr. 9, 2020, Laurent Pharmaceuticals announced that a spin off from McGill University planned to test its…

On Apr. 9, 2020, James Petrie, a graduate student in applied mathematics at the University of Waterloo in…
On Apr. 9, 2020, Codex DNA announced a newly developed and released suite of products and services designed…

On Apr. 9, 2020, BioSig Technologies announced that an article titled “The IMPDH inhibitor merimepodib suppresses SARS-COV-2 replications”…

On Apr. 9, 2020, Washington University School of Medicine in St. Louis launched a clinical trial for patients…

On Apr. 9, 2020, LabCorp and Ciox Health announced an agreement to collaborate on a comprehensive U.S.-based COVID-19…

On Apr. 9, 2020, BioCryst Pharmaceuticals announced the company has opened enrollment into a randomized, double-blind, placebo-controlled clinical…

On Apr. 9, 2020, Pfizer and BioNTech disclosed additional details of their collaboration to advance candidates from BioNTech’s…

On Apr. 9, 2020, Chembio Diagnostics has been selected for use in a Stony Brook Medicine effort to…

On Apr. 9, 2020, the U.S. Department of Agriculture (USDA) confirmed the presence of highly pathogenic H7N3 avian…
On Apr. 9, 2020, Dr. Carl Wesolowski at the University of Saskatchewan discovered that metformin, often used to…

On Apr. 9, 2020, iBio announced the signing of two agreements with the Infectious Disease Research Institute (IDRI)…
On Apr. 9, 2020, CytoDyn announced a patient with severe COVID-19 under the care of a leading medical…

On Apr. 9, 2020, Pfizer announced that the FDA had approved BRAFTOVIᆴ (encorafenib) in combination with cetuximab (marketed…

On Apr. 8, 2020, Chembio Diagnostics announced a second $1.5 million purchase order from UNICEF for multiplex Zika,…

On Apr. 8, 2020, CSL Behring and SAB Biotherapeutics announced a partnership to combat the coronavirus pandemic with…