FDA issued Emergency Use Authorization for potential COVID-19 treatment
On May 1, 2020, the FDA issued an Emergency Use Authorization (EUA) for the investigational antiviral drug remdesivir…

On May 1, 2020, the FDA issued an Emergency Use Authorization (EUA) for the investigational antiviral drug remdesivir…
On May 1, 2020, a University of Southern California (USC) study suggested that temporarily suppressing the body’s immune…

On May 1, 2020, the CDC launched the National Healthcare Safety Network (NHSN) COVID-19 Module Data Dashboard showing…

On May 1, 2020, the U.S. Dept. of Veterans Affairs (VA) announced participation in a series of clinical…

On May 1, 2020, the CDC launched the SARS-CoV-2 Sequencing for Public Health Emergency Response, Epidemiology and Surveillance…

On May 1, 2020, CHF Solutions announced that it has shipped Aquadex product to a specialty care center…

On May 1, 2020, people with cancer who develop COVID-19 are much more likely to die from the…

On May 1, 2020, Athersys and University Hospitals Cleveland Medical Center (UH Cleveland) announced that UH Cleveland is…

On May 1, 2020, XBiotech announced that human immune donors had been identified that can support the Company’s…
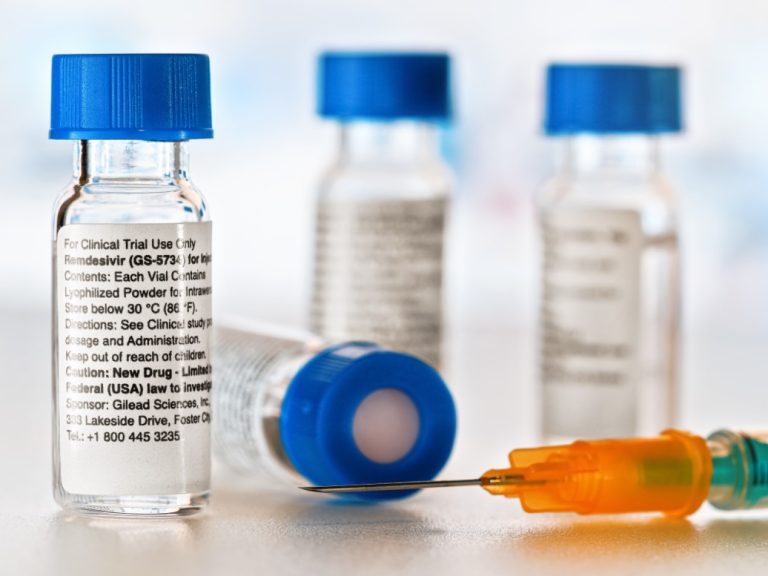
On May 1, 2020, Gilead announced the FDA granted emergency use authorization (EUA) for the investigational antiviral remdesivir…

On Apr. 30, 2020, the FDA included, under the ventilator emergency use authorization (EUA), a ventilator developed by…

On Apr. 30, 2020, Mesoblast announced a Phase 2/3 randomized, placebo-controlled trial to rigorously confirm whether its allogeneic…

On Apr. 30, 2020, AstraZeneca and the University of Oxford announced an agreement for the global development and…

On Apr. 30, 2020, University of Oxford announced an agreement with the UK-based global biopharmaceutical company AstraZeneca for…
On Apr. 30, 2020, FUJIFILM announced that it will reserve manufacturing capacity for a future COVID-19 therapy for…
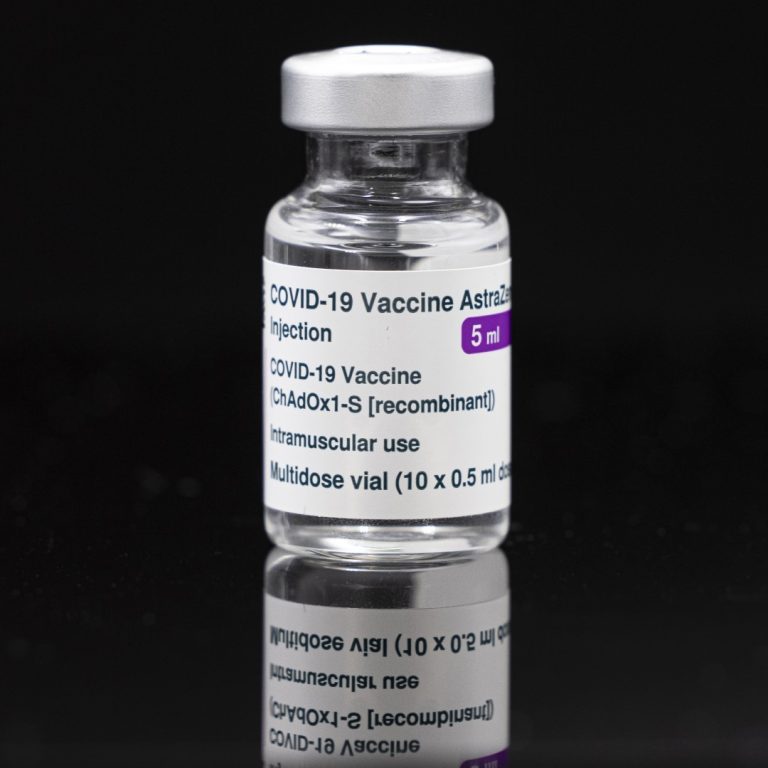
On Apr. 30, 2020, the University of Oxford announced an agreement with the UK-based global biopharmaceutical company AstraZeneca…

On Apr. 30, 2020, The California Institute for Regenerative Medicine (CIRM) awarded $750,000 to City of Hopeメs John…

On Apr. 30, 2020, Vaxart announced it has obtained positive pre-clinical results for its COVID-19 vaccine candidates, with…

On Apr. 30, 2020, Mateon Therapeutics reported several peer-reviewed publications derived from its R&D effort against COVID-19. Published…

On Apr. 30, 2020, Eiger BioPharmaceuticals announced that the first patients have been dosed in a Phase 2…
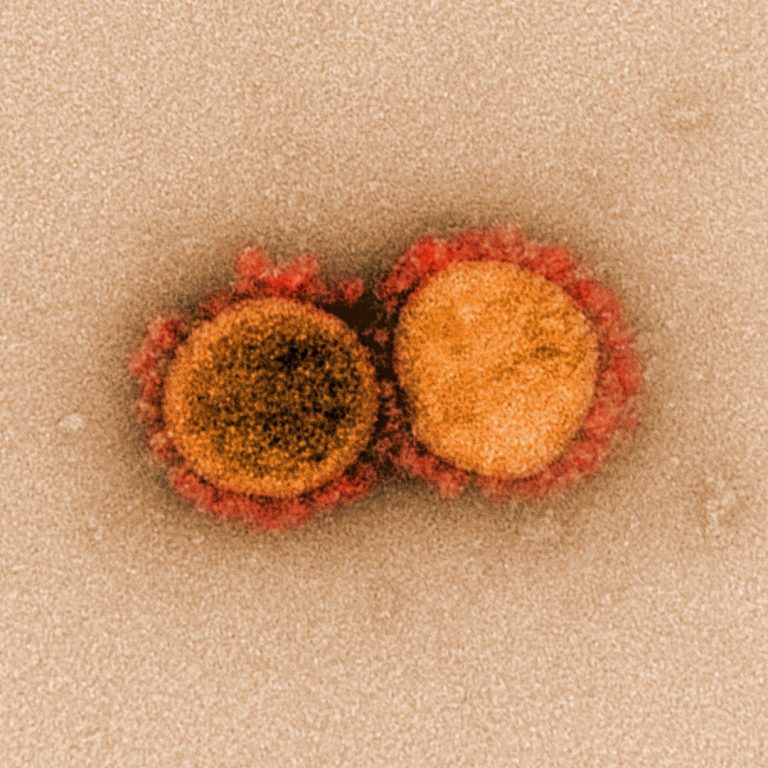
On Apr. 30, 2020, an international team of more than 120 scientists has detailed the impact of 75…
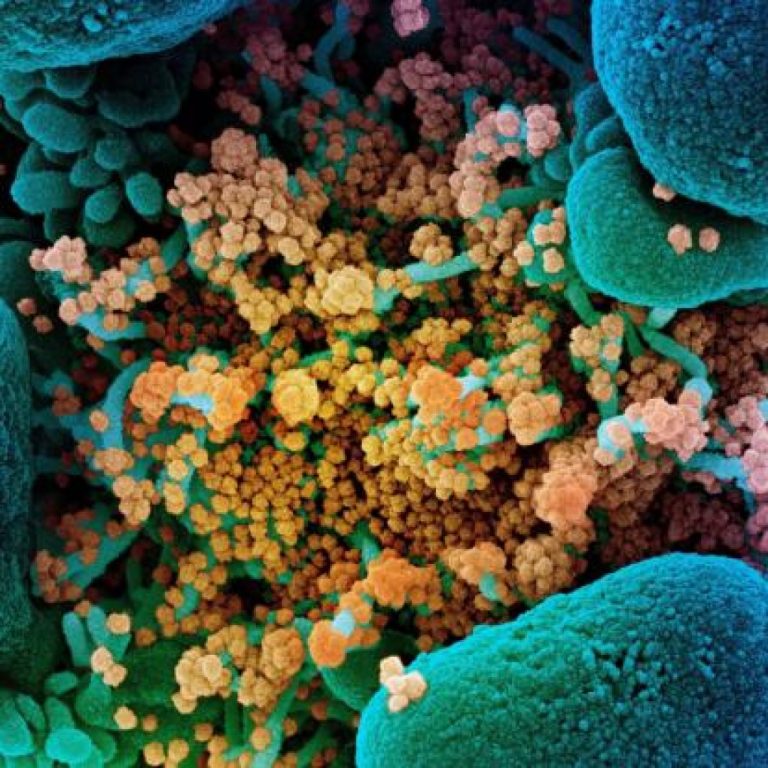
On Apr. 30, 2020, Emory University researchers are taking part in a multi-site study across the U.S. to…

On Apr. 30, 2020, INOVIO announced an agreement to expand its manufacturing partnership with the German contract manufacturer…

On Apr. 29, 2020, the U.S. Dep. of Veterans Affairs (VA) announced a partnership with the XPRIZE Foundationメs…

On Apr. 29, 2020, the NIH announced a new initiative aimed at speeding innovation, development and commercialization of…

On Apr. 29, 2020, Gilead announced topline results from the open-label, Phase 3 SIMPLE trial evaluating 5-day and…

On Apr. 29, 2020, an anti-inflammatory drug developed at Scripps Research 25 years ago tested as a way…
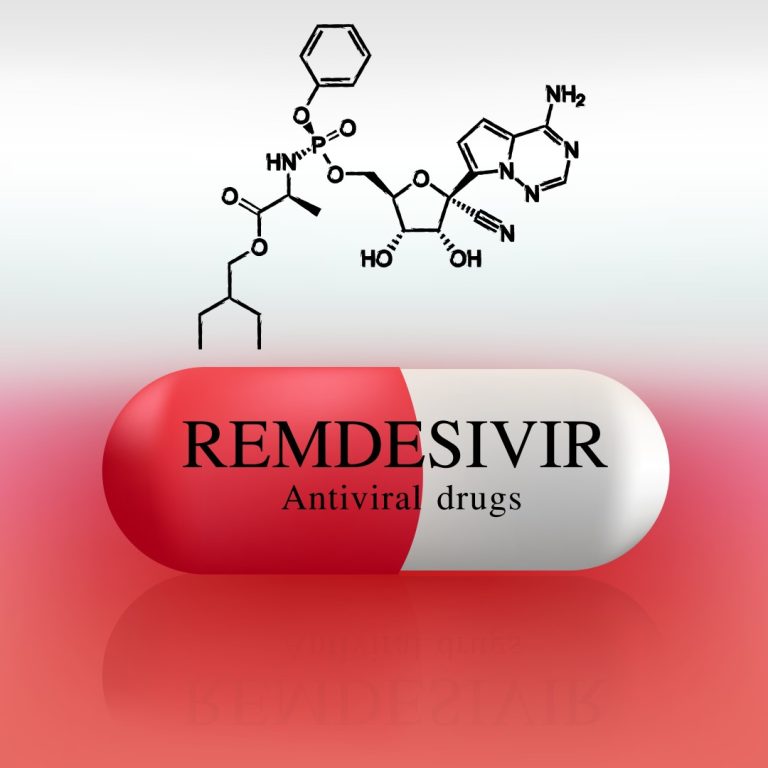
On Apr. 29, 2020, Emory University played a leading role in the government-sponsored clinical trial of the COVID-19…

On Apr. 29, 2020, hospitalized patients with advanced COVID-19 and lung involvement who received remdesivir recovered faster than…

On Apr. 29, 2020, Catalent announced a collaboration with the Janssen Pharmaceutical Companies of Johnson & Johnson, whereby…