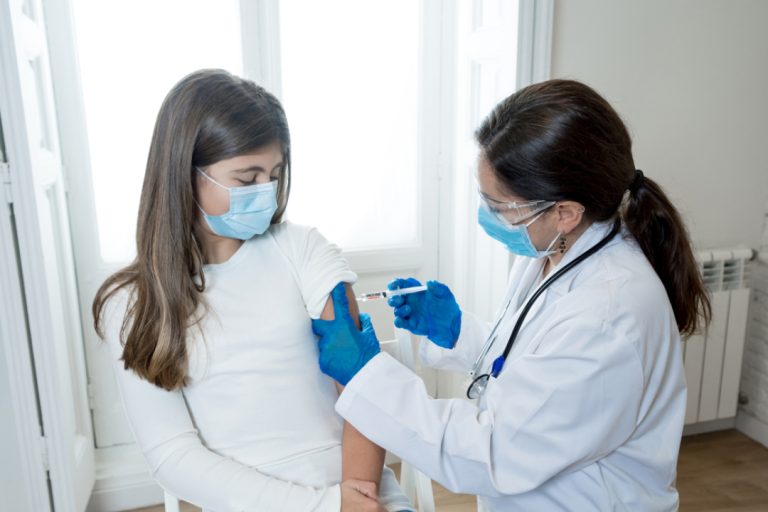
BioNTech and Fosun Pharma announced regulatory approval of mono- and bivalent COVID-19 vaccine COMIRNATYᆴ in individuals 12 years and older in Hong Kong
On Dec. 23, 2022, BioNTech and Fosun Pharmaceutical announced that they had received the certificates of registration as…