OraSure Technologies receives three EUA’s for its COVID-19 rapid antigen tests for non-prescription use
On Jun. 7, 2021, OraSure Technologies announced that it had received Emergency Use Authorization (EUA) from the FDA…

On Jun. 7, 2021, OraSure Technologies announced that it had received Emergency Use Authorization (EUA) from the FDA…

On Jun. 3, 2021, Resverlogix announced a partnership to support planned commercialization of apabetalone in the United States,…

On Jun. 3, 2021, the National Institutes of Health announced that the experimental drug TEMPOL may be a…

On Jun. 3, 2021, Innovation Pharma announced that it had achieved full patient enrollment in its randomized, double-blind,…

On Jun. 3, 2021, Moder’s vaccine against COVID-19, to support the government’s ongoing efforts to secure access to…

On Jun. 2, 2021, RELIEF THERAPEUTICS reported that its collaboration partner, NRx Pharmaceuticals, announced that it had submitted…

On Jun. 2, 2021, Moderna and UNICEF announced that they had signed a long-term agreement for the supply…

On Jun. 2, 2021, Vaxxas announced that research demonstrated the potential of Vaxxasメ novel high-density microarray patch (HD-MAP)…

On Jun. 2, 2021, Lonza announced the expansion of its collaboration with Moderna. The expanded collaboration extended the…

On Jun. 1, 2021, a study by researchers at the National Institutes of Health and their colleagues found…

On Jun. 1, 2021, WHO validated the Sinovac-CoronaVac COVID-19 vaccine for emergency use, giving countries, funders, procuring agencies…

On Jun. 1, 2021, the National Institutes of Health announced that it had started a Phase 1/2 clinical…

On Jun. 1, 2021, the WHO announced it had validated the Sinovac-CoronaVac COVID-19 vaccine for emergency use, giving…

On Jun. 1, 2021, Moderna announced that it had initiated the rolling submission process with the U.S. Food…

On Jun. 1, 2021, Moderna announced that it had entered into an agreement with Thermo Fisher Scientific for…

On Jun. 1, 2021, the U.S. Department of Health and Human Services (HHS) unveiled a new type of…

On Jun. 1, 2021, CytoDyn announced that Chiral Pharma in the Philippines placed its first purchase order for…

On May 28, 2021, Humanigen announced that the company had submitted an application to the FDA requesting Emergency…

On May 28, 2021, Omeros announced preliminary results from the second cohort of critically ill COVID-19 patients treated…

On May 27, 2021, Novartis and Molecular Partners announced the start of the clinical trial EMPATHY, a Phase…

On May 27, 2021, Innovation Pharma announced that patient enrollment in the Company’s 120-patient, Phase 2 clinical trial…

On May 27, 2021, Innovation Pharmaceuticals announced that patient enrollment in the Companyメs 120-patient, Phase 2 clinical trial…
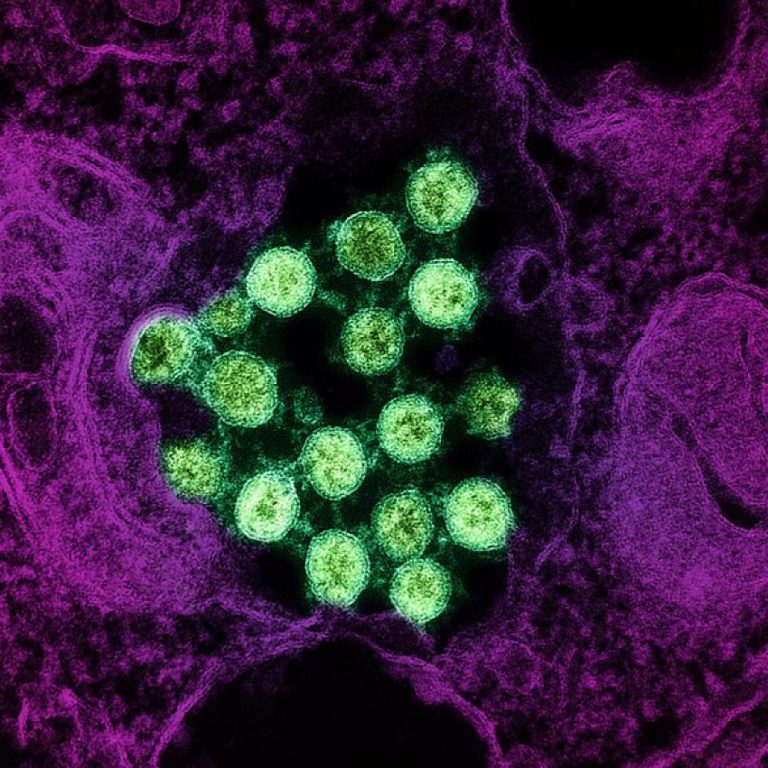
On May 27, 2021, Seattle Children’s researchers published a study that uncovered a deeper understanding of why people…

On May 27, 2021, Sanofi and GlaxoSmithKline started enrolment in their Phase 3 clinical study to assess the…

On May 26, 2021, Henry Schein announced the Company was awarded a $53.4 million contract from the U.S….

On May 26, 2021, the National Center for Biotechnology Information announced a report that aimed to analyze the…

On May 26, 2021, GlaxoSmithKline and Vir Biotechnology announced the U.S. Food and Drug Administration (FDA) granted an…

On May 25, 2021, Moderna announced that the Phase 2/3 study of its COVID-19 vaccine (mRNA-1273) in adolescents…

On May 25, 2021, ImmunityBio announced two South African studies to examine the potential for using its hAd5…
Cast of Characters: Donald Trump, President | Mike Pence, Vice President | Anthony S. Fauci, MD, Director, National…