Emergent BioSolutions awarded 10-Year BARDA contract valued at $704 million for Ebanga treatment for Ebola
On Jul. 31, 2023, Emergent BioSolutions announced that it was awarded a 10-year contract by the Biomedical Advanced…
On Jul. 31, 2023, Emergent BioSolutions announced that it was awarded a 10-year contract by the Biomedical Advanced…
On Jul. 28, 2023, the U.S. Food and Drug Administration (FDA) approved Merck’s Ervebo, a vaccine for the…

On Apr. 17, 2023, the World Health Organization (WHO) announced that six additional laboratory-confirmed cases of Marburg virus…

On Mar. 21, 2023, the Ministry of Health of the United Republic of Tanzania declared an outbreak of…
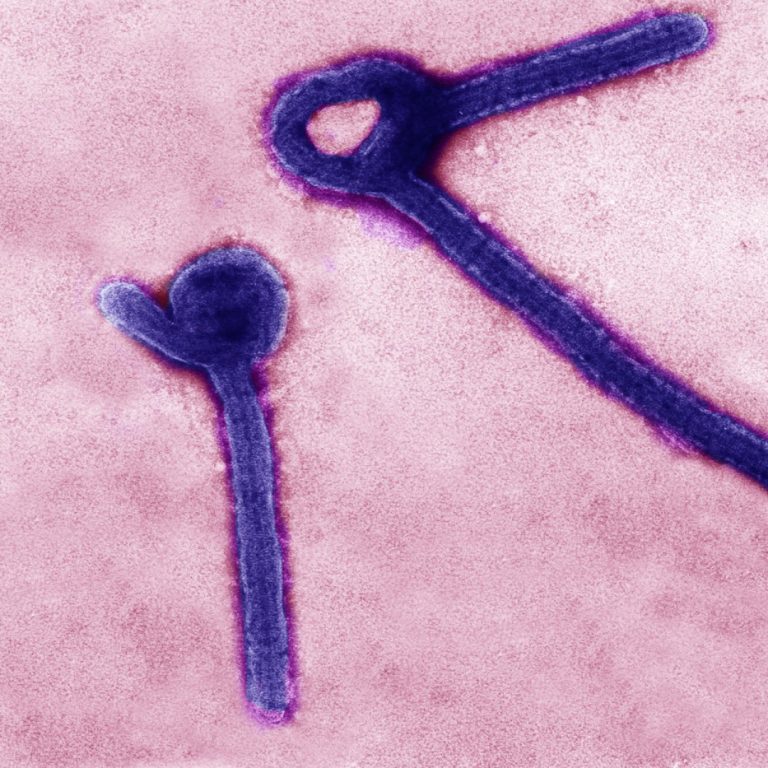
On Feb. 2, 2023, a National Institutes of Health research group with extensive experience studying ebolavirus countermeasures successfully…
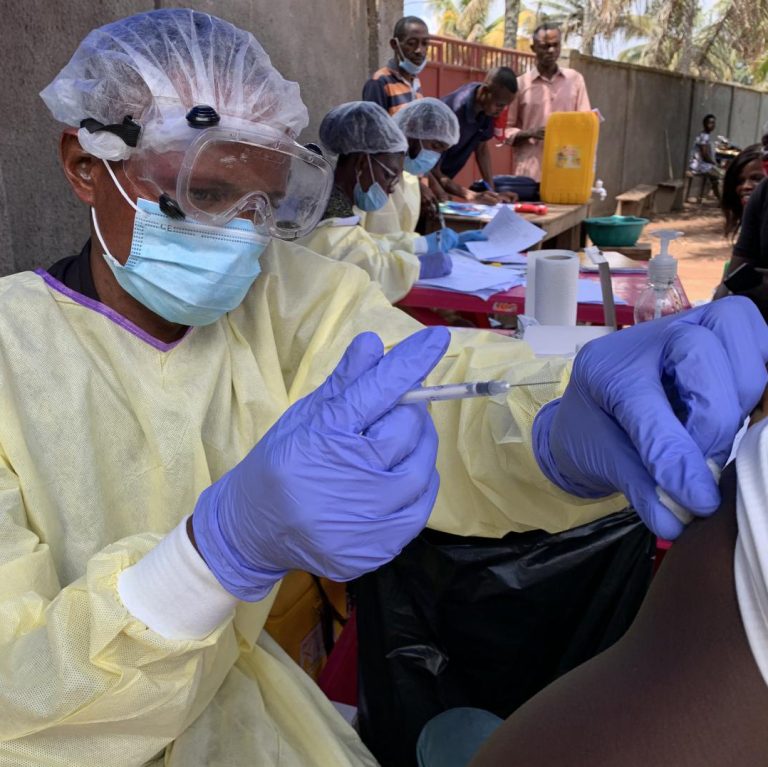
On Jan. 11, 2023, Uganda declared the end of the Ebola disease outbreak caused by Sudan ebolavirus, less…
On Dec. 15, 2022, the University of Oxford’s Ebola vaccine candidate has been shipped to Uganda, just 80…

On Dec. 9, 2022, the World Health Organization (WHO) announced that the first doses of one of the…
On Sept. 20, 2022, The health authorities in Uganda declared an outbreak of Ebola after a case of…
On Sept. 17, 2022, the Democratic Republic of the Congo declared the end of an Ebola outbreak that…

On Apr. 28, 2022, the Ministry of Health of the Democratic Republic of the Congo declared an outbreak…

On Dec. 16, 2021, the Ebola outbreak that erupted in the Democratic Republic of the Congo’s North Kivu…

On Aug. 14, 2021, the Ministry of Health of Cote d’Ivoire confirmed the country’s first case of Ebola…
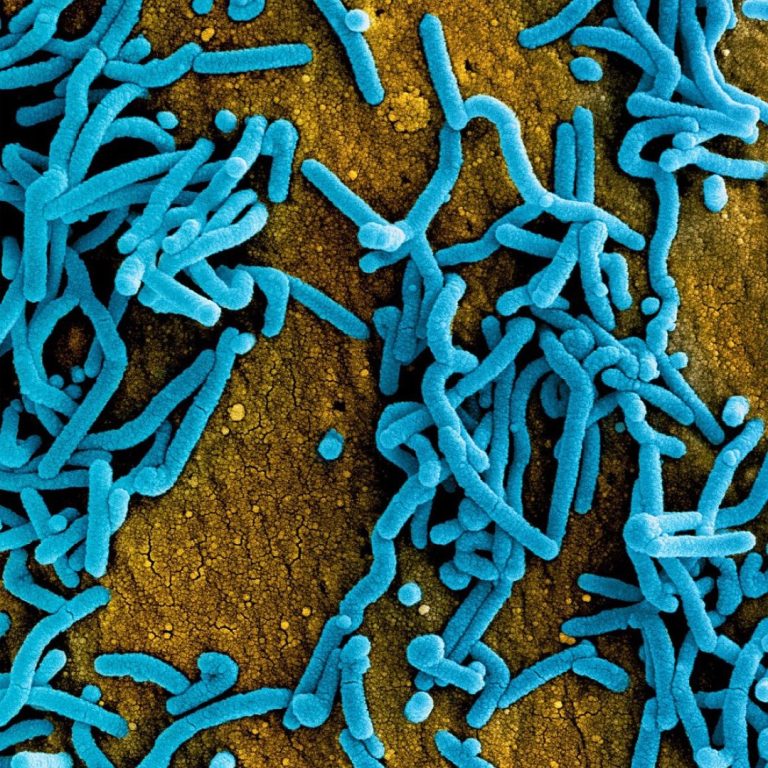
On Aug. 9, 2021, health authorities in Guinea confirmed a case of Marburg virus disease in the southern…

On Jun. 19, 2021, the Ebola outbreak that emerged in Guinea in mid-February was declared over. It was…
On May 3, 2021, the World Health Organization (WHO) announced the end of the 12th Ebola outbreak in…

On Apr. 19, 2021, Tonix Pharmaceuticals and OyaGen announced an exclusive worldwide licensing agreement for an antiviral inhibitor…

On Feb. 14, 2021, the WHO announced that Health authorities in Guinea had declared an outbreak of Ebola…

On Dec. 21, 2020, the U.S. National Institutes of Health (NIH) announced a study published in the Proceedings…
On Oct. 14, 2020, Regeneron announced that the FDA had approved Inmazeb (atoltivimab, maftivimab and odesivimab-ebgn) for the…
On Oct. 14, 2020, the FDA approved Regeneron Pharmaceutical’s Inmazeb (atoltivimab, maftivimab, and odesivimab-ebgn), a mixture of three…
On Aug. 26, 2020, researchers announced an inexpensive, sensitive, CRISPR-based tests diagnose two deadly viruses in settings with…

On Jul. 29, 2020, Regeneron announced that the Biomedical Advanced Research and Development Authority (BARDA) had entered into…
On May 29, 2020, Johnson & Johnson announced that its Janssen Pharmaceutical subsidiary received a positive opinion from…

On Apr. 30, 2020, BioSig Technologies announced that an article titled, ‘The IMPDH inhibitor merimepodib has similar antiviral…
On Apr. 16, 2020, Regeneron announced the U.S. Food and Drug Administration (FDA) had accepted for Priority Review…
On Mar. 23, 2020, Soligenix announced that its ongoing collaboration with the University of Hawaii at Manoa (UH…

On Mar. 17, 2020, CanSino Biologics announced that its recombinant novel Coronavirus Vaccine (Adenovirus Type 5 Vector) candidate…
On Mar. 9, 2020, researchers from the National Institute of Allergy and Infectious Diseases (NIAID), part of the…

On Mar. 20, 2018, in response to the Ebola outbreak that claimed more than 10,000 lives in West…