New Genetic Risk Score Better Predicts Diabetes, Obesity and Downstream Complications
On Mar. 16, 2026, investigators from Mass General Brigham announce they have built metabolic polygenic risk scores (PRS) for predicting…

On Mar. 16, 2026, investigators from Mass General Brigham announce they have built metabolic polygenic risk scores (PRS) for predicting…

On Jan. 5, 2026, Novo Nordisk announced that the Wegovy® pill is now available, providing those seeking help…

On Dec. 22, 2025, Novo Nordisk announced announced that the US Food and Drug Administration (FDA) has approved…

On Dec. 3, 2025, Massachusetts Institute of Technology (MIT) engineers announced they can accurately measure blood glucose by…

On Nov. 18, 2025, Stanford Medicine researchers reported that a combination blood stem cell and pancreatic islet cell…

On Nov. 6, 2025, the Institute for Health Metrics and Evaluation (IHME) at the University of Washington School…

On Nov. 5, 2025, a large study of adults with type 2 diabetes showed that those who consistently…

On Oct. 27, 2025, researchers from Johns Hopkins Medicine and the Johns Hopkins Bloomberg School of Public Health…

On Oct. 5, 2025, Eli Lilly ill invest more than $1 billion in India in the coming years…

On Sept. 25, 2025, Biolinq, a pioneering developer of precision multi-analyte biosensors to improve metabolic health, announced the…
On Sept. 17, 2025, The New England Journal of Medicine published the results from the OASIS 4 phase…

On Sept. 8, 2025, the Institute for Health Metrics and Evaluation (IHME) at the University of Washington School…

On Sept. 5, 2025, in a medical first, researchers report that they have implanted CRISPR-edited pancreas cells into…

On Aug. 20, 2025, the National Institute for Health and Care Excellence (NICE) announced that millions of people…

On Aug. 18, 2025, Novo Nordisk announced the launch of a new offer that enables self-paying, eligible, type…

On Jul. 9, 2025, MIT engineers announced they have designed an implantable reservoir that can remain under the…

On Jun. 20, 2025, Vertex Pharmaceuticals announced publication of updated data from the Phase 1/2 portion of the…
On Jun. 16, 2025, the National Institute for Health and Care Excellence (NICE) announced that the recommendation in…

On May 12, 2025, Roche announced it plans to invest up to $550 million in its Diagnostics site…

On Mar. 3, 2025, the Institute for Health Metrics and Evaluation reported that without urgent policy reform and…

On Feb. 21, 2025, Novo Nordisk announced that the U.S. Food and Drug Administration (FDA) has determined the…
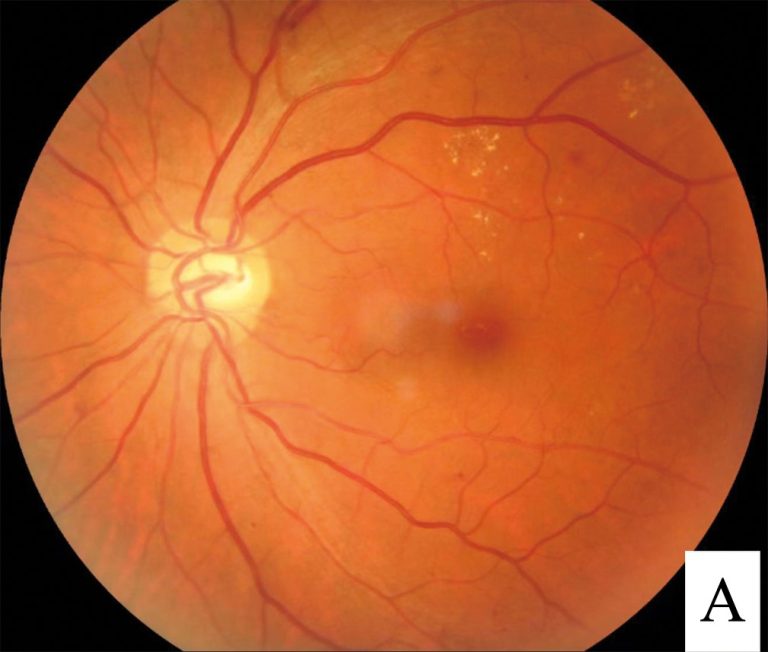
On Feb. 4, 2025, Genentech announced that the U.S. Food and Drug Administration (FDA) had approved Susvimo® (ranibizumab…

On Jan. 28, 2025, Novo Nordisk announced that the U.S. Food and Drug Administration (FDA) has approved Ozempic® to…

On Jan. 20, 2025, an analysis of data from nearly 2 million people has revealed new insights into…
On Jan. 13, 2025, a study led by NYU Langone Health shows that the risk of developing dementia at…

On Dec. 20, 2024, the U.S. Food and Drug Administration (FDA) approved Eli Lilly’s Zepbound (tirzepatide) for the…

On Dec. 19, 2024, the U.S. Food and Drug Administration announced it had updated the definition of “healthy…

On Nov. 15, 2024, the AARP, the Alzheimer’s Disease Data Initiative (AD Data Initiative), and the Institute for…

On Nov. 13, 2024, the NCD Risk Factor Collaboration (NCD-RisC) with support from the World Health Organization (WHO)…
On Nov. 4, 2024, a team of University of Wisconsin–Madison researchers warned that artificial intelligence tools gaining popularity…