Sorrento announced discovery of salicyn-30 small molecule that strongly inhibits SARS-CoV-2 infection
On Oct. 7, 2020, Sorrento Therapeutics announced that it had discovered a small molecule termed Salicyn-30 that demonstrated…
On Oct. 7, 2020, Sorrento Therapeutics announced that it had discovered a small molecule termed Salicyn-30 that demonstrated…

On Oct. 7, 2020, Eli Lilly announced additional details on its SARS-CoV-2 neutralizing antibody programs ヨ including interim…
On Sept. 30, 2020, ViralClear Pharmaceuticals and Sorrento Therapeutics announced the companies were exploring the synergistic potential of…

On Sept. 23, 2020, the FDA issued an emergency use authorization (EUA) for the first serology (antibody) point-of-care…
On Sept. 22, 2020, Oxford Immunotec announced that it had entered into an extensive research collaboration with Public…
On Sept. 21, 2020, XBiotech announced it was developing a new breakthrough candidate therapy it calls FLUVID for…

On Sept. 18, 2020, Roche announced the launch of its Elecsysᆴ Anti-SARS-CoV-2 S antibody test for markets accepting…

On Sept. 17, 2020, Eli Lilly and Amgen announced a global antibody manufacturing collaboration to significantly increase the supply…
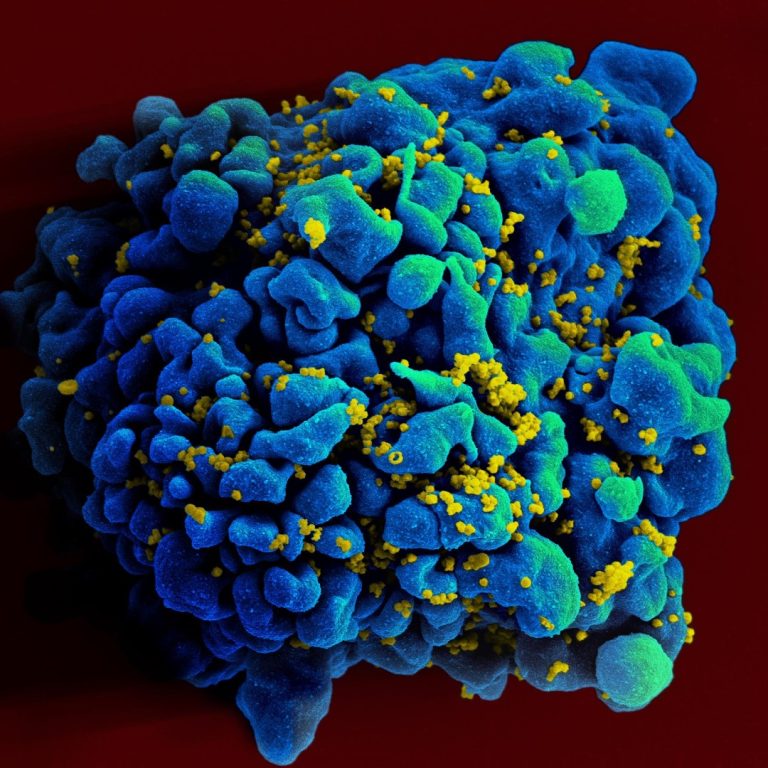
On Sept. 16, 2020, a study by researchers at La Jolla Institute for Immunology published in the Cell,…
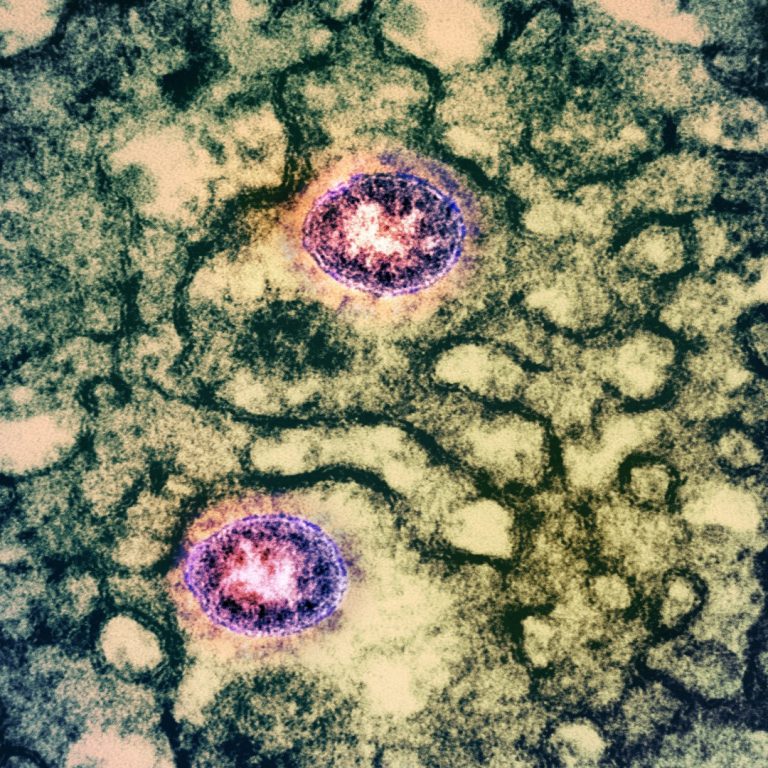
On Sept. 16, 2020, AXIM Biotechnologies announced that it had filed an Emergency Use Authorization (EUA) application with…
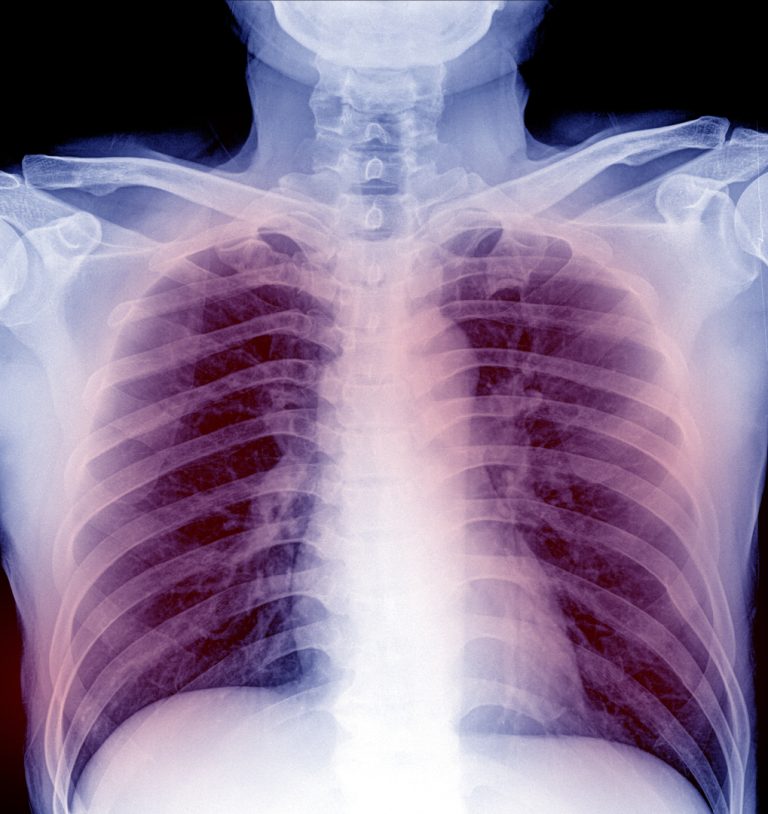
On Sept. 15, 2020, the University of Texas Health Science Center at Houston (UTHealth) announced that a study…

On Sept. 14, 2020, Meridian Bioscience announced the launch of a novel highly sensitive SARS-CoV-2 antibody pair to…

On Sept. 10, 2020, Corvus Pharmaceuticals announced updated data from its ongoing Phase 1 study investigating the potential…

On Sept. 3, 2020, ADMA Biologics announced the launch of COVID-19 ImmunoRank Neutralization MICRO-ELISA, a proprietary, fully-validated ELISA…

On Sept. 1, 2020, scientists at deCODE genetics in Iceland, a subsidiary of Amgen, published a study in…

On Aug. 25, 2020, AstraZeneca announced that the first participants have been dosed in a Phase I trial…

On Aug. 24, 2020, AXIM Biotechnologies announced that it had signed an exclusive limited licensing, manufacturing and distribution…

On Aug. 24, 2020, XBiotech announced that the U.S. Food and Drug Administration (FDA) issued an emergency use…
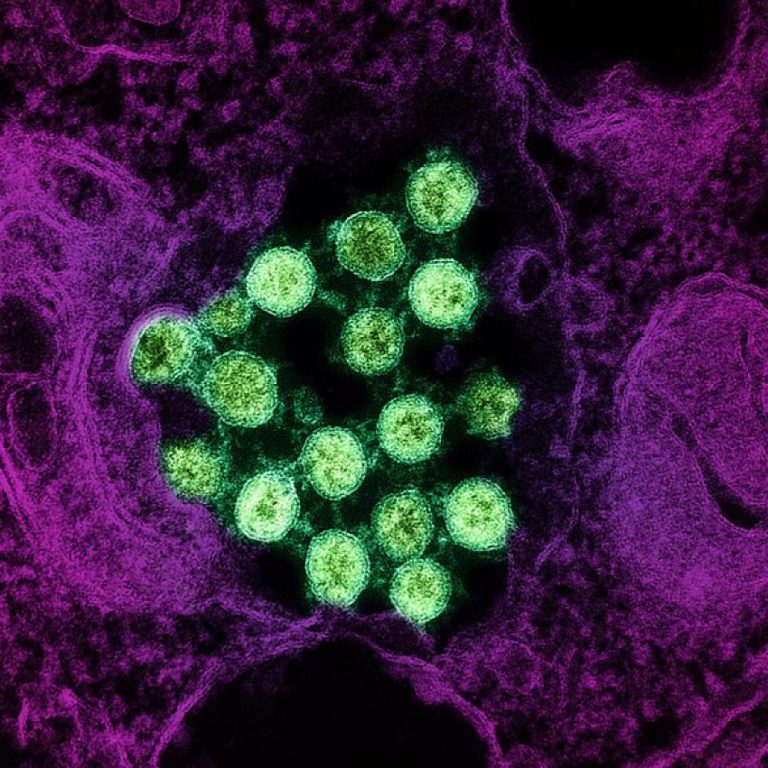
On Aug. 20, 2020, the National Institute of Allergy and Infectious Diseases (NIAID), one of the National Institutes…

On Aug. 19, 2020, Accelerate Diagnostics and BioCheck announced that the FDA had issued an Emergency Use Authorization…
On Aug. 17, 2020, Scientisfic American reported that Costa Rican scientists at the Clodomiro Picado Institute have inoculated…

On Aug. 17, 2020, XBiotech announced that it had identified True Human antibodies that could potentially be used…

On Aug. 14, 2020, Medigen Vaccine Biologics (MBV) published preclinical results of their COVID-19 vaccine candidate in BioRxiv,…

On Aug. 13, 2020, Heat Biologics reported preclinical data for Heat’s gp96-based COVID-19. The data, generated at the…

On Aug. 11, 2020, LabCorpᆴ announced details of a no charge antibody testing program in response to federal…

On Aug. 10, 2020, two Phase 3, randomized, placebo-controlled, double-blind clinical trials testing whether experimental monoclonal antibodies (mAbs)…

On Aug. 10, 2020, iBio provided an update on one of its proprietary vaccine candidates being designed to…

On Aug. 5, 2020, AXIM Biotechnologies announced the development, patent filing and Emergency Use Approval (EUA) filing of…

On Aug. 4, 2020, Precipio announced it had completed the internal validation of the U.S. Food and Drug…

On Jul. 30, 2020, Johnson & Johnson announced that its lead vaccine candidate protected against infection with SARS-CoV-2,…