Federal Medicare rule required all long-term care facilities to offer vaccination for influenza and pneumococcal disease
On Oct. 7, 2005, a new Federal Medicare rule became effective that required all long-term care facilities to…

On Oct. 7, 2005, a new Federal Medicare rule became effective that required all long-term care facilities to…
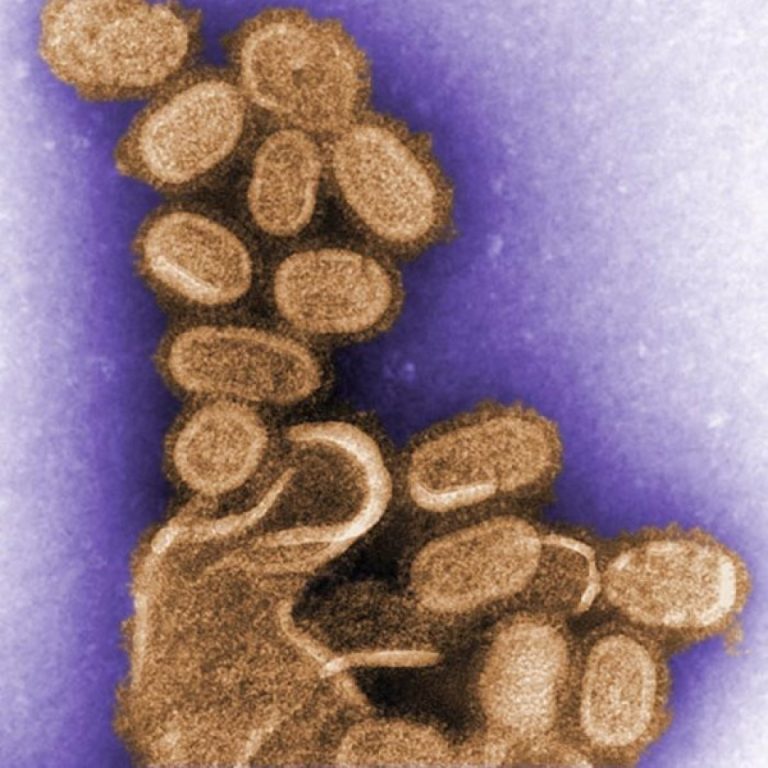
On Oct. 7, 2005, Jeffery Taubenberger, AH Reid, AE Krafft, Karen Bijwaard and Thomas Fanning published a report…

On Sept. 6, 2005, the vaccine that combined the measles, mumps, rubella, and varicella antigens (Proquad by Merck)…
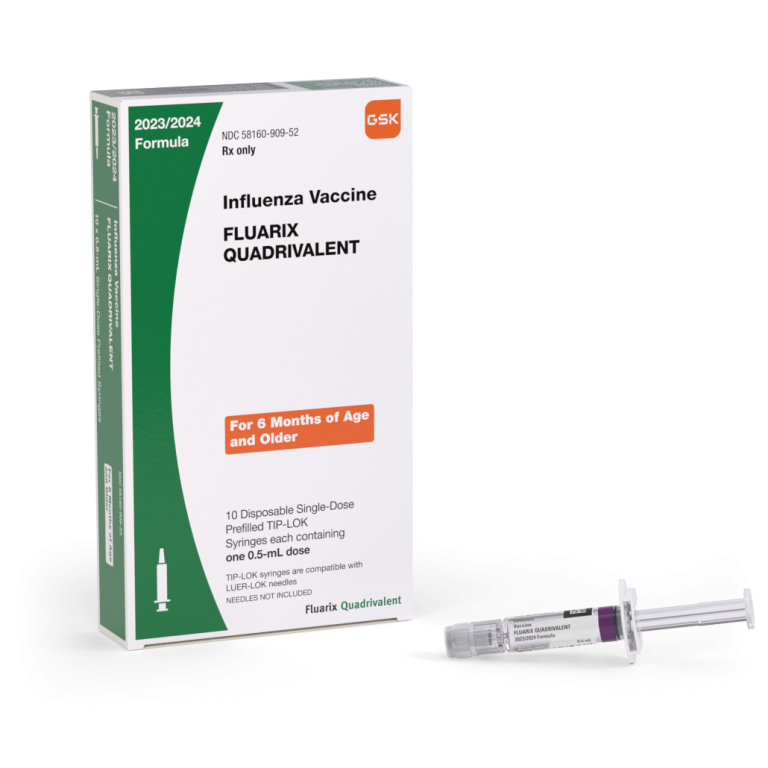
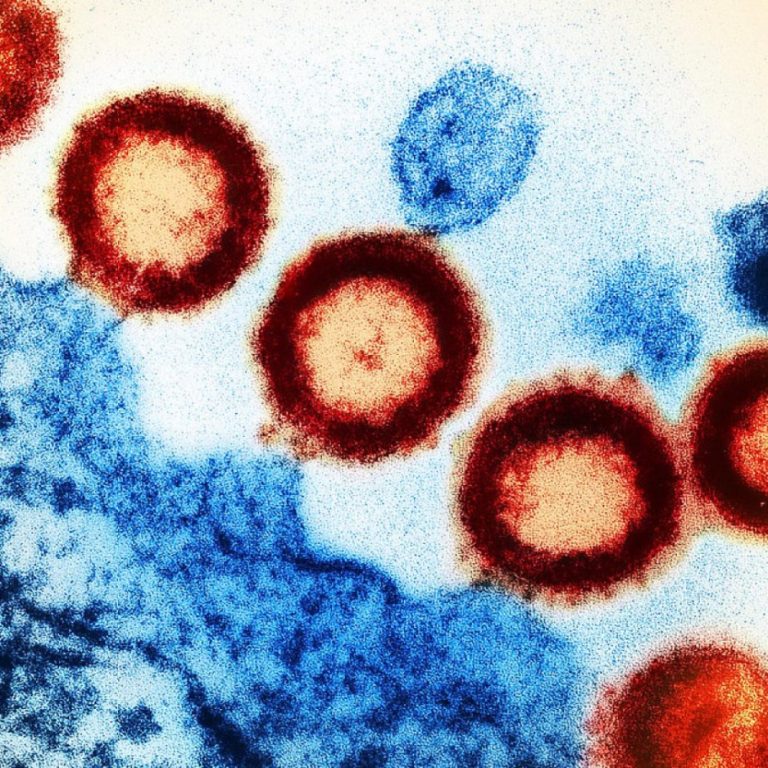
On Aug. 31, 2005, the inactivated, injectable influenza vaccine (Fluarix) by GlaxoSmithKline (GSK) was licensed. The vaccine was…
On Aug. 11, 2005, the Food and Drug Administration (FDA) approved an application of a pediatric/adolescent formulation of…

On Jul. 12, 2005, Corixa Corp., based on First Hill in Seattle, was acquired by Glaxosmithkline (GSK) for…
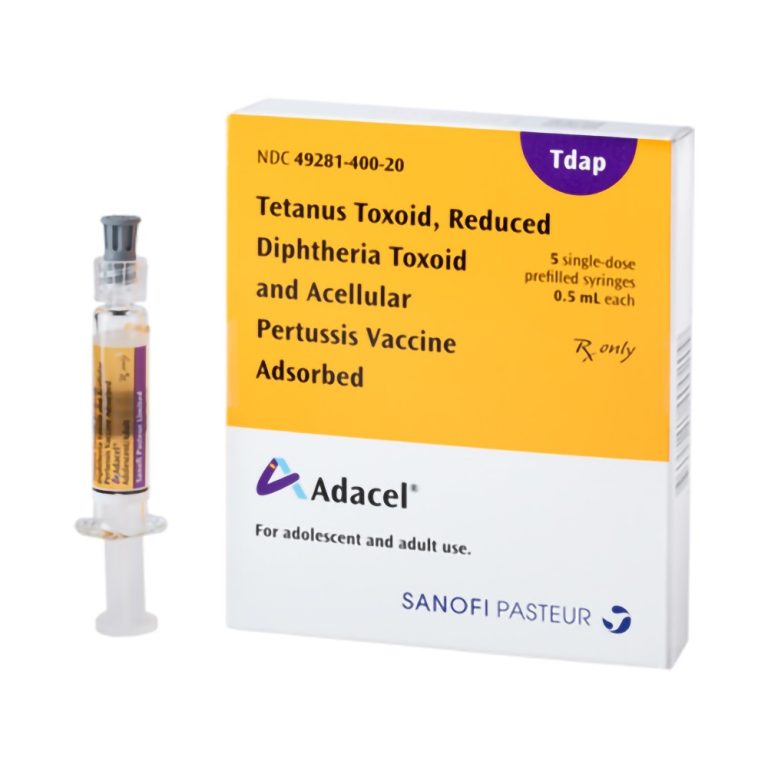
On Jun. 9, 2005, the U.S. Food and Drug Administration (FDA) licensed a 2nd Tdap vaccine (Adacel by…
On May 3, 2005, the cellular pertussis vaccine combined with the adult formulation of tetanus and diphtheria (Tdap:…
On Apr. 22, 2005, Sanofi Pasteur announced the U.S. Food and Drug Administration (FDA) had granted licensure to…
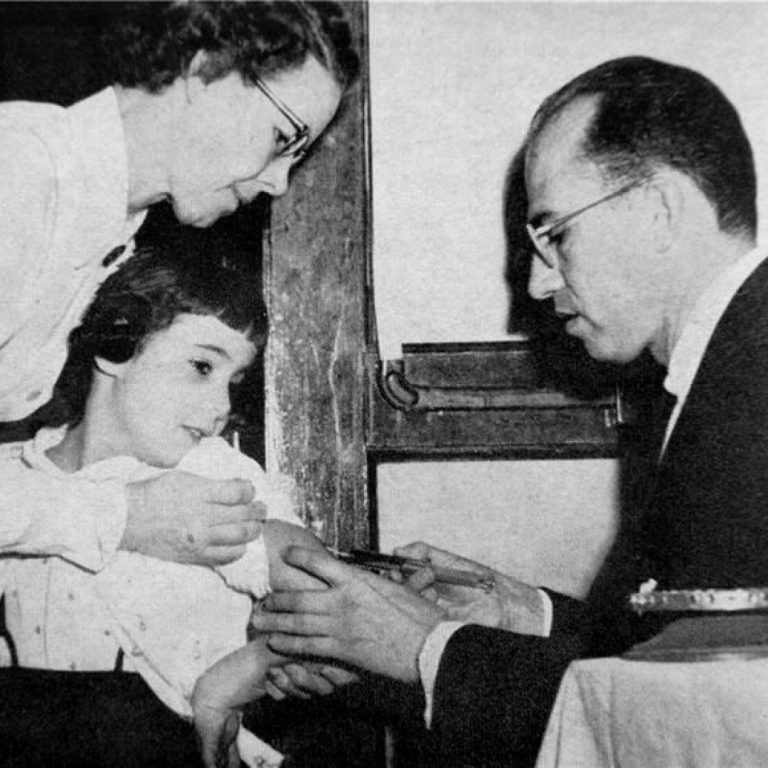
On Apr. 12, 2005 marked the 50th anniversary of the announcement that the polio vaccine, developed by Jonas…
On Apr. 1, 2005, Sanofi pasteur was awarded a five-year $97 million contract from the U.S. Department of…
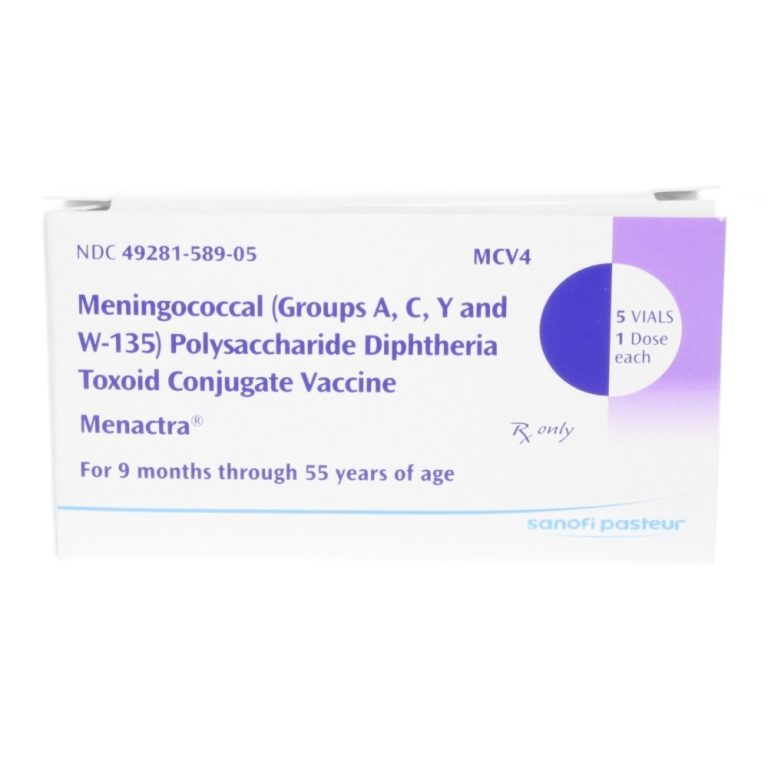
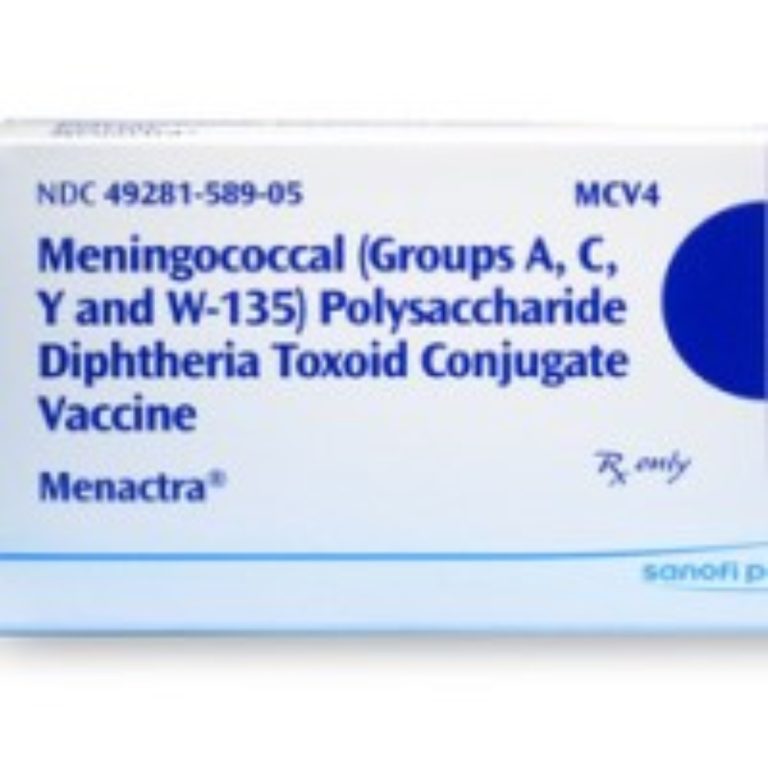
On Jan. 14, 2005, the first meningococcal polysaccharide (Serogroups A, C, Y and W-135) diphtheria toxoid conjugate vaccine…
On Nov. 1, 2004, an Oregon Health & Science University (OHSU) research team announced that it was one…

On Sept. 14, 2004, researchers at OHSU and OHSU’s Vaccine and Gene Therapy Institute announced a collaboration with…

On Aug. 25, 2004, a significant shortage of influenza vaccine occurred in the U.S. as a result of…
On Jun. 26, 2004, the 8th and final report of the Immunization Safety Review Committee was issued by…

On May 27, 2004, the National Institute of Allergy and Infectious Diseases (NIAID) announced it had awarded contracts…
On May 25, 2004, researchers at the Vaccine and Gene Therapy Institute (VGTI) and the Oregon National Primate…
On May 4, 2004, the National Institute of Allergy and Infectious Diseases (NIAID), awarded a new license agreement…

On Mar. 1, 2004, the National incident Management System (NIMS), developed by the Department of Homeland Security (DHS),…

In 2004, the University of Georgia Cancer Center was established, specializing in drug targets, diagnostic tests, cancer vaccines,…
In 2004, Rubella, also known as German measles, was declared eliminated in the U.S. Before the rubella vaccination…

On Oct. 15, 2003, the Immunization Practices Advisory Committee (ACIP) voted to recommend that children 6 to 23…
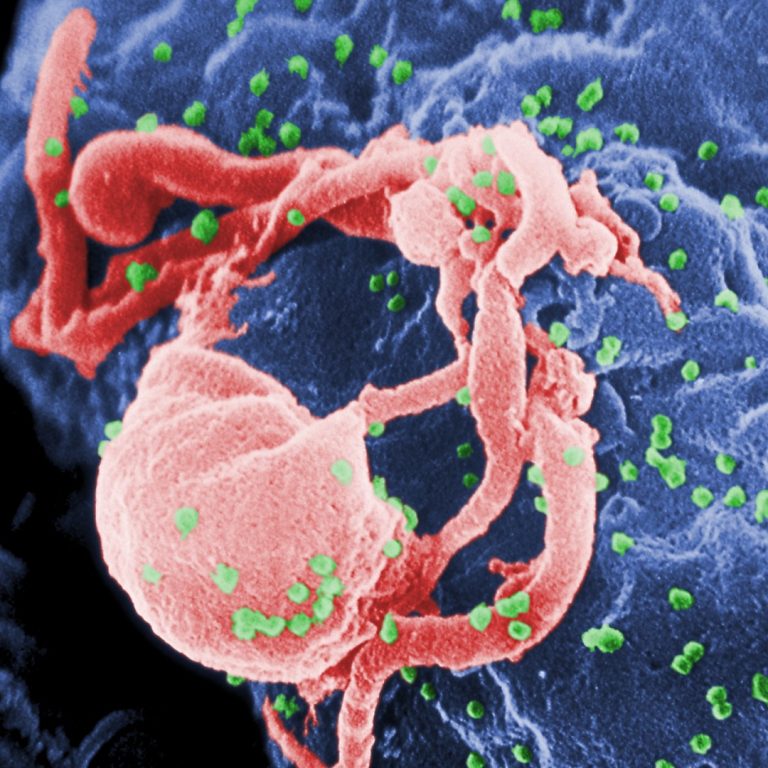
On Sept. 29, 2003, $81 million was awarded by National Institute of Allergy and Infectious Diseases (NIAID) through…
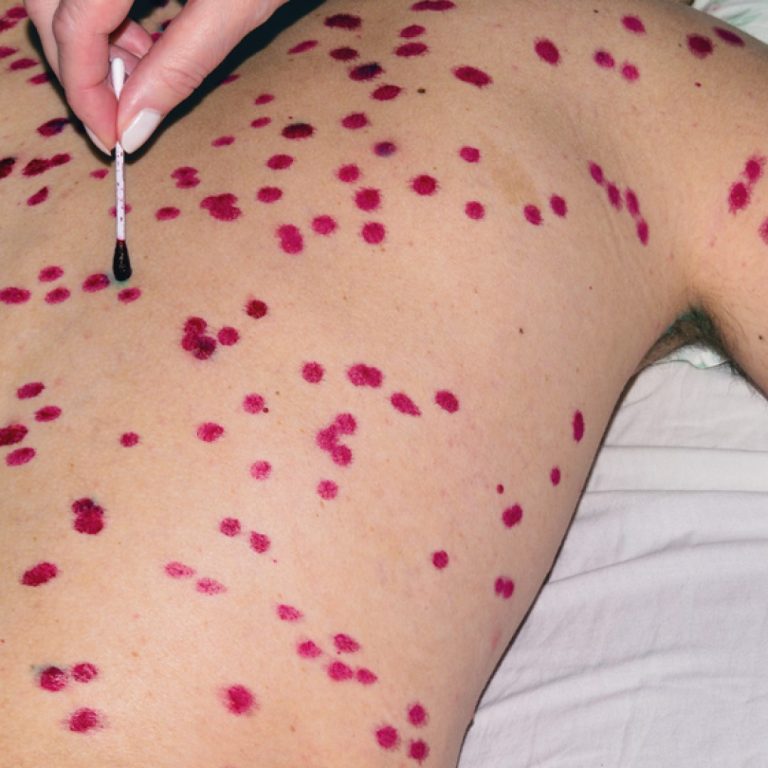
On Aug. 17, 2003, Oregon Health & Science University (OHSU) announced the final results of a smallpox vaccine…

On Jul. 8, 2003, the U.S. Centers for Disease Control and Prevention (CDC) responded to U.S. outbreak of…

On Jun. 17, 2003, the first nasally administered influenza vaccine (FluMist by MedImmune) was licensed. This live influenza…

On Apr. 14, 2003, researchers at Canada’s Michael Smith Genome Science Centre in Vancouver announced they had sequenced…

On Mar. 18, 2003, Severe Acute Respiratory Syndrome (SARS) was placed on U.S. quarantine list. Persons under quarantine…
On February 24 2003, VaxGen announced that AIDSVAX B/B did not prove effective in the trials conducted in…