Johnson & Johnson COVID-19 booster showed substantial increase in antibody and T-cell responses
On Dec. 5, 2021, Johnson & Johnson announced preliminary results from an independent study, including a subset of…

On Dec. 5, 2021, Johnson & Johnson announced preliminary results from an independent study, including a subset of…

On Dec. 5, 2021, Sorrento Therapeutics announced the peer-reviewed publication of a series of novel SARS-CoV-2 MPro inhibitors…

On Dec. 3, 2021, Gavi, the Vaccine Alliance, approved a new malaria vaccination programme to support the introduction,…

On Dec. 3, 2021, a report from the U.S. Department of Health and Human Services (HHS) found that…

On Dec. 2, 2021, National Resilience and Harvard University announed they had established a five-year research and development…
On Dec. 1, 2021, T2 Biosystems announced that its T2SARS-CoV-2 Panel detected the Omicron COVID-19 variant (B.1.1.529). The…

On Dec. 1, 2021, Moderna announced a revised supply agreement with the UK government for up to 60…
On Dec. 1, 2021, SIGA Technologies announced that Health Canada had approved oral TPOXX (tecovirimat) as an extraordinary…
On Nov. 30, 2021, Inovio Pharma announced the company was rapidly moving to evaluate its COVID-19 DNA vaccine…
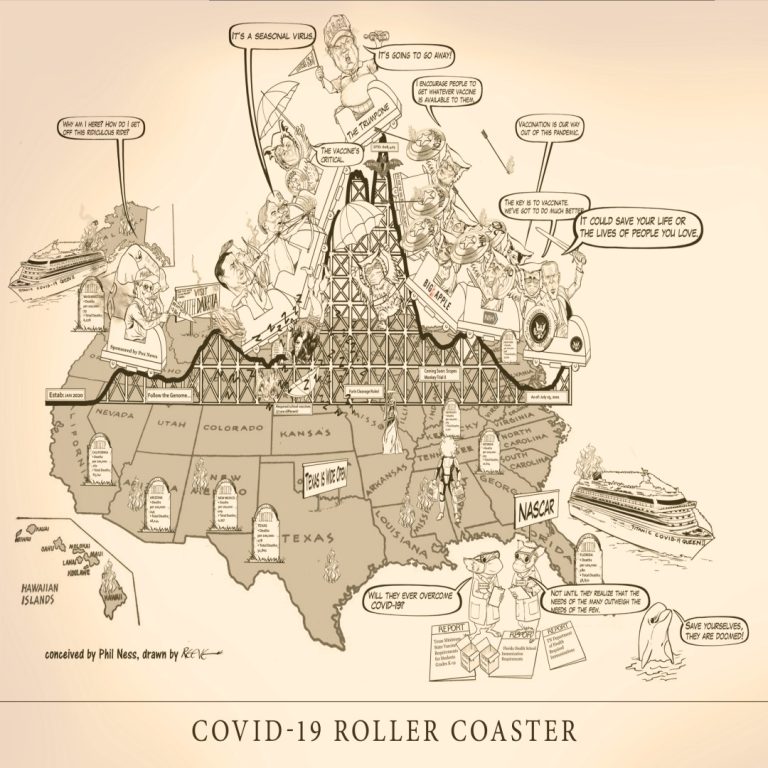
The COVID-19 Roller Coaster is a wild ride, strap yourself in and hold on… Cast of Characters: Senator…

On Nov. 26, 2021, Merck provided an update on the MOVe-OUT study of molnupiravir, an investigational oral antiviral…

On Nov. 25, 2021, Pfizer and BioNTech announced that the Committee for Medicinal Products for Human Use (CHMP)…

On Nov. 24, 2021, Johnson & Johnson announced the U.S. Food and Drug Administration (FDA) had issued Emergency…

On Nov. 24, 2021, Novavax announced its submission to the Singapore Health Sciences Authority for interim authorization of…

On Nov. 23, 2021, the World Health Organization’s COVID-19 Technology Access Pool and the Medicines Patent Pool finalized…
On Nov. 22, 2021, Pfizer announced topline results from a longer-term analysis of the safety and efficacy of…
On Nov. 21, 2021, Gavi, the Vaccine Alliance, announced that India has completed the national introduction of pneumococcal…

On Nov. 19, 2021, the U.S. Food and Drug Administration (FDA) announced that it had extended the emergency…

On Nov. 17, 2021, Novavax and and Serum Institute of India announced that the Philippine Food and Drug…

On Nov. 17, 2021, Novavax announced that the European Medicines Agency (EMA) had begun its evaluation of an…

On Nov. 17, 2021, Zosano Pharma announced that the Philippine Food and Drug Administration had granted emergency use…

On Nov. 17, 2021, GlaxoSmithKline and Vir Biotechnology announced U.S. government contracts totalling approximately $1 billion (USD) to…

On Nov. 16, 2021, an agreement was announced that enabled the European Union and European Economic Area countries…

On Nov. 16, 2021, the University of Oxfordメs and AstraZeneca announced that two billion doses of the ChAdOx1…

On Nov. 15, 2021, AstraZeneca reported that 2 billion doses of the COVID-19 vaccine had been released for…

On Nov. 15, 2021, Moderna confirmed that Health Canada had authorized the use of a booster dose of…

On Nov. 15, 2021, Novavax and SK bioscience announced submission of a Biologics License Application (BLA) for Novavax’…

On Nov. 11, 2021, the University of Oxford began recruiting for a Phase I trial to test an…

On Nov. 11, 2021, Zipline, Pfizer and BioNTech announced that Zipline had successfully completed the first long-range drone…

On Nov. 10, 2021, the World Health Organization (WHO) announced that while reported measles cases had fallen compared…