Ivermectin ineffective in large study of treatment among patients with Covid-19
On Mar. 31, 2022, researchers in Brazil reported that a study of COVID-19 patients at 12 clinics that…

On Mar. 31, 2022, researchers in Brazil reported that a study of COVID-19 patients at 12 clinics that…

On Mar. 31, 2022, Sorrento Therapeutics announced that the FDA had given clearance to commence the Phase 3…

On Mar. 31, 2022, researchers announced they have created a complete, gap-free sequence of the roughly 3 billion…

On Mar. 30, 2022, Lucira Health announced that preliminary clinical trial results for its candidate COVID-19 & Flu…
On Mar. 29, 2022, Pfizer and BioNTech announced that the U.S. Food and Drug Administration (FDA) had expanded…

On Mar. 22, 2022, Pfizer announced an agreement with UNICEF to supply up to 4 million treatment courses…

On Mar. 21, 2022, Twist Bioscience announced the launch of specific synthetic RNA positive controls for SARS-CoV-2 encapsulated…

On Mar. 17, 2022, the National Institutes of Health announced that nearly 100,000 highly diverse whole genome sequences…
On Mar. 17, 2022, Soligenix announced the results of a booster vaccination study using CiVax™ (heat stable COVID-19…

On Mar. 10, 2022, Cocrystal Pharma announced dosing of the first subjects in a Phase 1 clinical trial…

On Mar. 10, 2022, schools with mandatory masking during the Delta surge had approximately 72% fewer cases of…

On Mar. 9, 2022, Pfizer announced that it had initiated a Phase 2/3 study, EPIC-PEDS (Evaluation of Protease…

On Mar. 4, 2022, the U.S. Centers for Disease Control and Prevention (CDC) reported that COVID-19 vaccination coverage with…

On Mar. 3, 2022, the World Health Organization (WHO) and the Medicines Patent Pool (MPP) jointly welcomed the announcement…

On Mar. 3, 2022, a study carried out in southwestern Ontario identified a highly mutated variant of SARS-CoV-2…

On Mar. 2, 2022, Roche announced an initial donation of essential medicines to Ukraine. Roche is working diligently…

On Mar. 2, 2022, the Iowa Department of Agriculture and Land Stewardship and the United States Department of…
On Mar. 2, 2022, Sorrento Therapeutics announced that its Phase I study of intranasal (IN) STI-9199 (COVISHIELDTM IN) has…

On Feb. 28, 2022, the Patent and Trial Appeal Board of the U.S. Patent and Trademark Office (USPTO)…
On Feb. 22, 2022, Sorrento Therapeutics announced that additional preclinical results demonstrate broad spectrum COVISHIELD (STI-9167) neutralizing activity…

On Feb. 1, 2022, University of Virginia Cancer Center announced it had been awarded a Comprehensive Cancer Center…

On Feb. 17, 2022, after studying blood samples from 244 patients hospitalized for COVID-19, a group of researchers,…

On Feb. 17, 2022, Resverlogix announced that the first Brazilian site had been initiated for its Phase 2b…
On Feb. 15, 2022, a woman with HIV who received a cord blood stem cell transplant to treat…
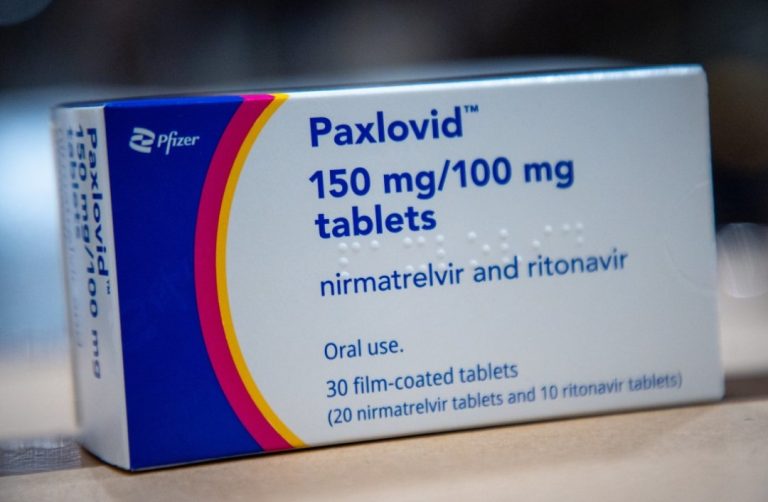
On Feb. 12, 2022, China’s medical products regulator announced that it had given conditional approval for Pfizer’s COVID-19…
On Feb. 11, 2022, Gilead Sciences announced new data from an interim analysis of its ongoing, Phase 2/3…

On Feb. 11, 2022, Gilead Sciences announced new data from an interim analysis of its ongoing, Phase 2/3…

On Feb. 8, 2022, Merck and Ridgeback Biotherapeutics announced that a total of 3.1 million courses of molnupiravir,…
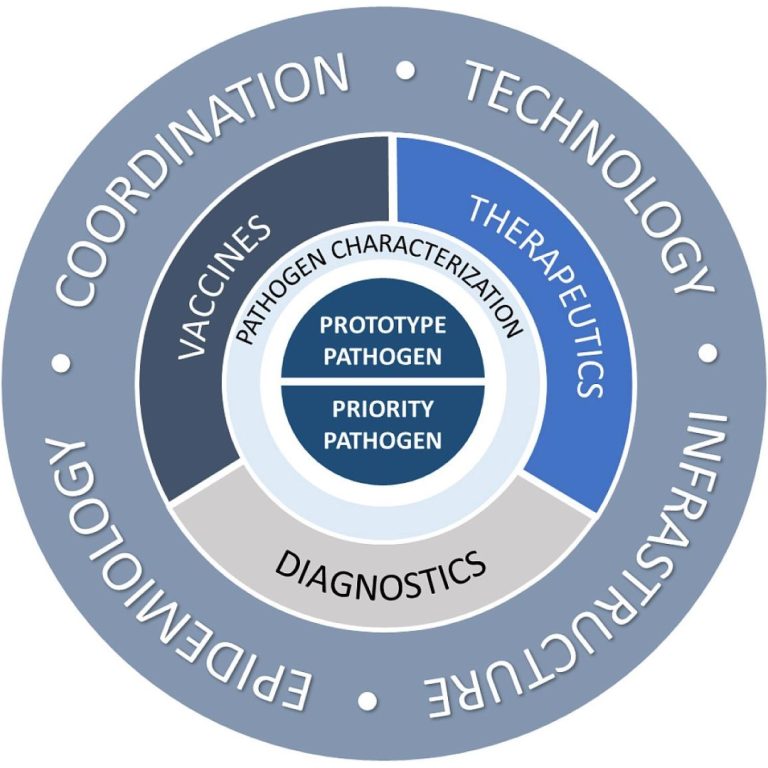
On Feb. 2, 2022, NIAID scientists announced the new Pandemic Preparedness Plan aimed to support critical basic and…

On Feb. 2, 2022, a new analysis of the first two patients treated in a clinical trial with…