NIH researchers identified potential new antiviral drug for COVID-19
On Jun. 3, 2021, the National Institutes of Health announced that the experimental drug TEMPOL may be a…

On Jun. 3, 2021, the National Institutes of Health announced that the experimental drug TEMPOL may be a…

On Jun. 1, 2021, the U.S. Department of Health and Human Services (HHS) unveiled a new type of…
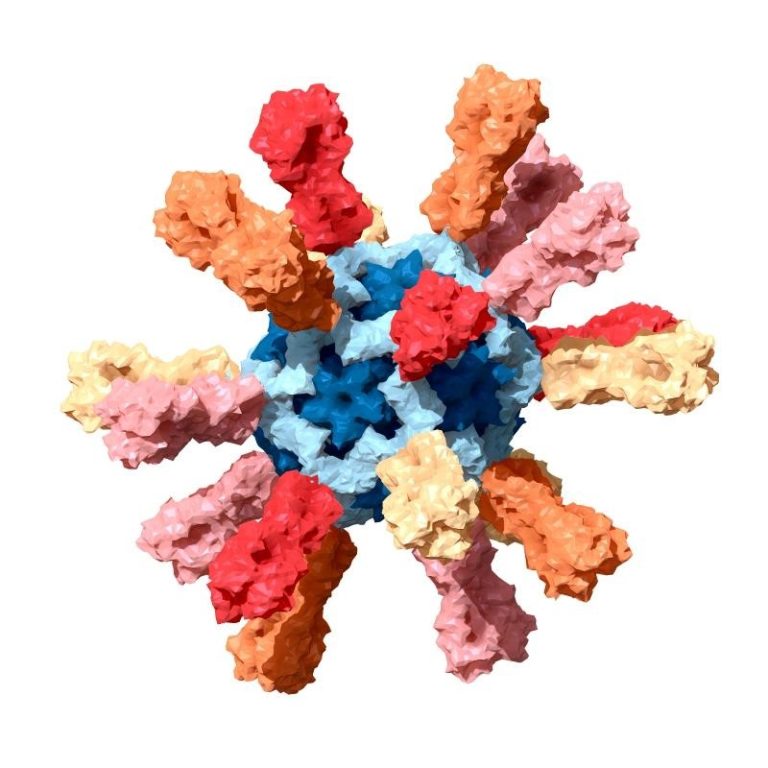
On Jun. 1, 2021, the National Institutes of Health (NIH) announced the first-in-human, Phase 1 trial assessing the…
On May 26, 2021, National Institutes of Health (NIH) scientists reported that the immune system’s attempt to eliminate…

On May 6, 2021, Trevena announced that TRV027, the Companyメs novel AT1 receptor selective agonist, had been selected…

On Apr. 29, 2021, the National Institutes of Health announced it was funding $29 million in additional grants…

COVID-19 and it’s naysayers are attacking Science and Reason. The defenders must suffer the slings and arrows from…

On Apr. 23, 2021, the NIH announced that a study assessing how people with immune system deficiencies or…
On Apr. 22, 2021, the NIH announced that a phase 3 trial to test the safety and efficacy…

On Apr. 21, 2021, the NIH announced that a Phase 2/3 trial to evaluate a new fully-human polyclonal…

On Apr. 19, 2021, the NIH announced that it was funding a large, randomized, placebo-controlled Phase 3 clinical…

On Apr. 15, 2021, the National Institute of Allergy and Infectious Diseases (NIAID) announced it had had established…
On Apr. 14, 2021, National Institutes of Health researchers reported that a study which compared to newborns conceived…

On Apr. 13, 2021, Rigel Pharma announced the completion of patient enrollment in a multi-center Phase 2 clinical…

On Apr. 9, 2021, Regeneron announced that newly updated National Institutes of Health (NIH) COVID-19 treatment guidelines strongly…

On Apr. 7, 2021, the National Institutes of Health (NIH) announced that a clinical trial was underway to…

On Apr. 7, 2021, Moderna announced publication of antibody persistence data out to 6 months following the second…

On Mar. 31, 2021, the National Institutes of Health (NIH) announced that an investigational vaccine designed to protect…
On Mar. 25, 2021, an international team of scientists announced they had found evidence that SARS-CoV-2 infects cells…

On Mar. 22, 2021, the National Institutes of Health (NIH) announced that results from a large clinical trial…

On Mar. 16, 2021, Moderna announced that the first participants had been dosed in the Phase 2/3 study,…

On Mar. 11, 2021, Rigel Pharma announced the completion of patient enrollment in a multi-center Phase 2 clinical trial to…
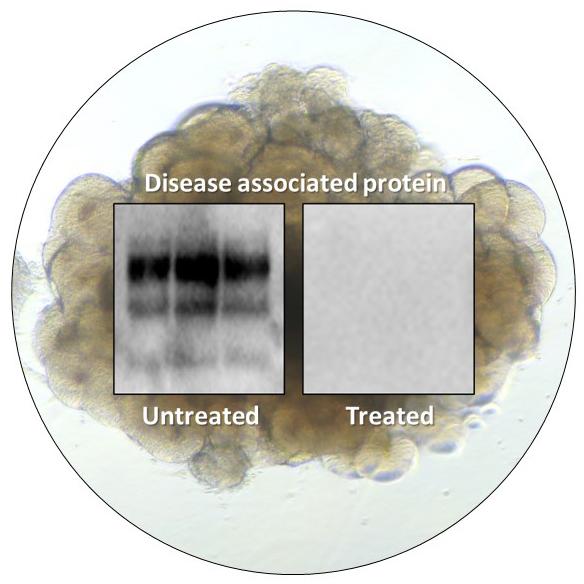
On Mar. 9, 2021, the National Institutes of Health (NIH) announced that approximately two years after establishing a…

On Mar. 8, 2021, the National Institutes of Health (NIH) announced that it had launched the last of…

On Mar. 4, 2021, the National Institutes of Health (NIH) announced that the ACTIV-3 clinical trial evaluating the…

On Mar. 2, 2021, the National Institutes of Health (NIH) announced it had launched the next version of…

On Mar. 2, 2021, the National Institutes of Health (NIH) halted a clinical trial evaluating the safety and…

On Mar. 2, 2021, the National Institutes of Health (NIH), announced that it had launched a research effort…

On Feb. 23, 2021, the NIH announced that a randomized, placebo-controlled Phase 1 clinical trial of two monoclonal…
On Feb. 17, 2021, the NIH annunced funding of a study to evaluate the effects of remdesivir in…