Pilot clinical trial in China to test University of British Columbia researcher’s findings as a targeted therapy for COVID-19
On Feb. 25, 2020, a University of British Columbia (UBC) researcher was part of an international team working…
On Feb. 25, 2020, a University of British Columbia (UBC) researcher was part of an international team working…
On Feb. 25, 2020, Vir Biotech and WuXi Biologics announced a development and manufacturing collaboration to advance and…
On Feb. 24, 2020, researchers at National Institute of Allergy and Infectious Disease’s Rocky Mountain Laboratories in Hamilton,…

On Feb. 24, 2020, Moderna announced it had released the first batch of mRNA-1273, the Company’s vaccine against…
On Feb. 24, 2020, Scientists at Texas Biomed began work on the novel coronavirus known as SARS-CoV-2. A…

On Feb. 24, 2020, Seqirus announced that the U.S. Food and Drug Administration (FDA) had approved the first…
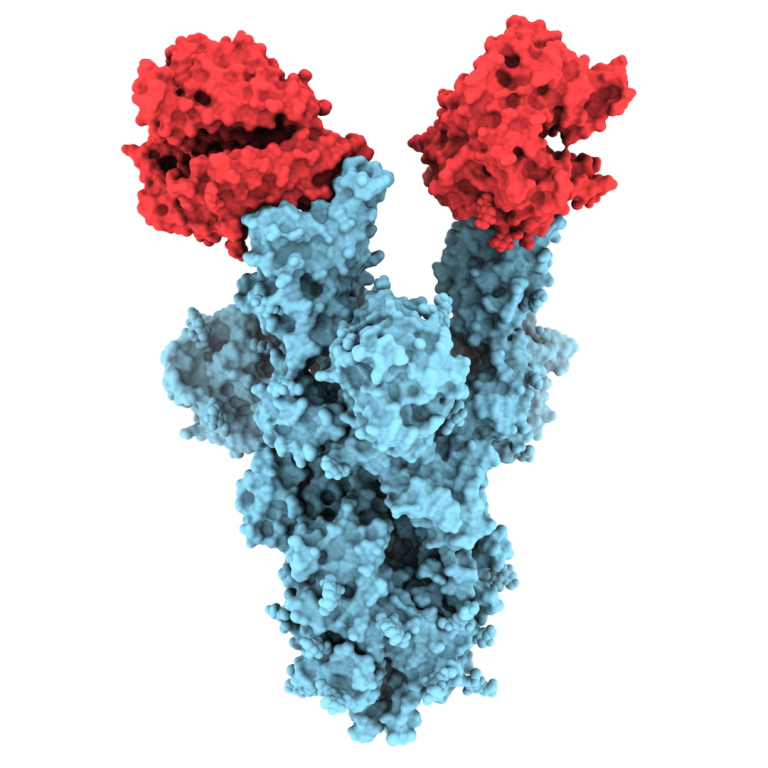
On Feb. 24, 2020, researchers reported that the spikes crowning the new coronavirus that causes COVID-19 atypical pneumonia…

On Feb. 19, 2020, NIAID scientists working with investigators from the University of Texas at Austin identified the…
On Feb. 18, 2020, Sanofi announced it had leveraged previous development work for a SARS vaccine which may…
On Jan. 29, 2020, Sorrento Therapeutics announced it had initiated a clinical and manufacturing collaboration with Celularity, a…

On Jan. 28, 2020, AbCellera confirmed that it had mobilized its pandemic response platform against the ongoing outbreak…
On Dec. 31, 2019, the World Health Organization (WHO) declared the Coronavirus outbreak a global pandemic. To date,…

On Dec. 11, 2019, Seqirus announced that its cell-based quadrivalent influenza vaccine (QIVc) had received approval from Health…

On Oct. 17, 2019, Genentech announced the U.S. Food and Drug Administration (FDA) had approved Xofluza (Baloxavir Marboxil),…
On Sept. 26, 2019, Seattle Children’s announced it was one of three recipients of $30 million in first-year-funding…
On Aug. 23, 2019, the Centers for Disease Control and Prevention (CDC) released Immunization Practices Advisory Committee (ACIP)…

On Apr. 24, 2019, GSK announced $100 million of new investment in its manufacturing site in Hamilton, Montana…

On Apr. 16, 2019, Sinovac Biotech announced that the positive results from the Company’s Phase III clinical study…
On Dec. 26, 2018, Sanofi announced the U.S. Food and Drug Administration (FDA) had approved VAXELIS (Diphtheria and…

On Dec. 5, 2018, Seqirus, a CSL subsidiary, announced real-world data showing that its cell-based quadrivalent influenza vaccine…

On Oct. 18, 2018, an international team of researchers reported that a surge in Lassa fever cases in…

On Oct. 3, 2018, Scripps Research announced that Calibr scientitsts from Scripp’s drug development division had begun using…

On Jun. 1, 2018, the American College of Obstetricians and Gynecologists (ACOG) issued a committee opinion on maternal…
On Apr. 3, 2018, the World Health Organization (WHO) recommended the introduction of typhoid conjugate vaccine (TCV) for…
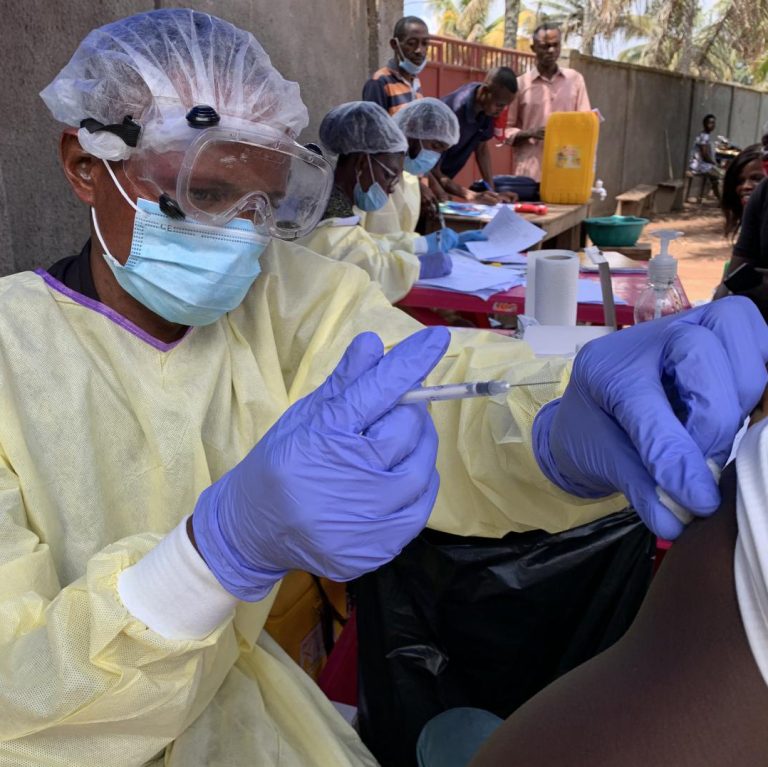
On Mar. 20, 2018, in response to the Ebola outbreak that claimed more than 10,000 lives in West…
On Jan. 12, 2018, the U.S. Centers for Disease Control and Prevention (CDC) published updated Immunization Practices Advisory…

On Jan. 9, 2018, armed with 1940s-vintage flu vaccine technology and supported by only anemic funding for developing…

On Aug. 7, 2017, Seqirus announced that the accelerated development of cell-based manufacturing technology at its state-of-the-art manufacturing…

On Jun. 15, 2017, the U.S. Department of Health and Human Services (HHS) published the nations’s updated Pandemic…

On April 26, 2017, the U.S. Department of Agriculture’s (USDA) announced that the New World screwworm (NWS) had…