U.S. Congress passed Medical Device Amendments to ensure safety and effectiveness of medical devices
In 1976, the U.S. Congress passed the Medical Device Amendments to ensure safety and effectiveness of medical devices,…

In 1976, the U.S. Congress passed the Medical Device Amendments to ensure safety and effectiveness of medical devices,…

In 1975, rapamycin, a macrolide produced by the bacterium Streptomyces hygroscopicus was first discovered and isolated from soil…

In Jul. 1974, In the Division of Pharmaceutical Service at the University of Iowa began producing cGMP compliant…

In 1974, the U.S. Food and Drug Administration (FDA) approved doxorubicin (Adriamycin), an antitumor anthracycline antibiotic from Streptomyces…

On Jun. 18, 1973, the U.S. Supreme Court upheld the 1962 drug effectiveness law and endorsed Food and…

On Feb. 17, 1973, the U.S. Food and Drug Administration (FDA) announced the recall of 29,500 institutional-size cans of…

On Oct. 27, 1972, the Consumer Product Safety Commission (CPSA) was enacted by the U.S. Congress. The CPSA…

On Jul. 1, 1972, the Regulation of Biologics–including serums, vaccines, and blood products–was transferred from the NIH to…

On May 11, 1972, the U.S. Food and Drug Administration (FDA) established Over-the-Counter (OTC) Drug Review began to enhance…

In 1972, the Division of Biologics Standards was transferred from the National Institutes of Health (NIH) to the U.S….
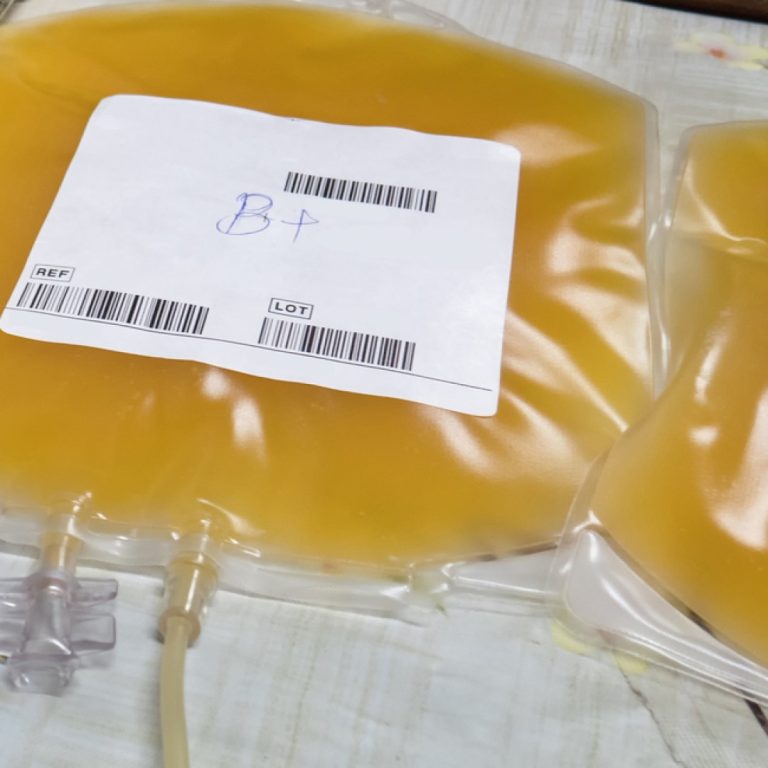
In 1972, the Food and Drug Administration’s (FDA) new Bureau of Biologics began to regulate all 7000 U.S….

On Sept. 29, 1971, the artificial sweetener saccharin, included in FDA’s original GRAS list, was removed from the…

On May 17, 1971, The Public Health Service’s Bureau of Radiological Health was transferred to the FDA.

On May 1, 1971, the Federal Comprehensive Drug Abuse Prevention and Control Act of 1970, more commonly known…

In 1971, the Public Health Service (PHS) Bureau of Radiological Health was transferred to U.S. Food and Drug…

On Feb. 27, 1970, in Upjohn v. Finch the Court of Appeals upheld enforcement of the 1962 drug…
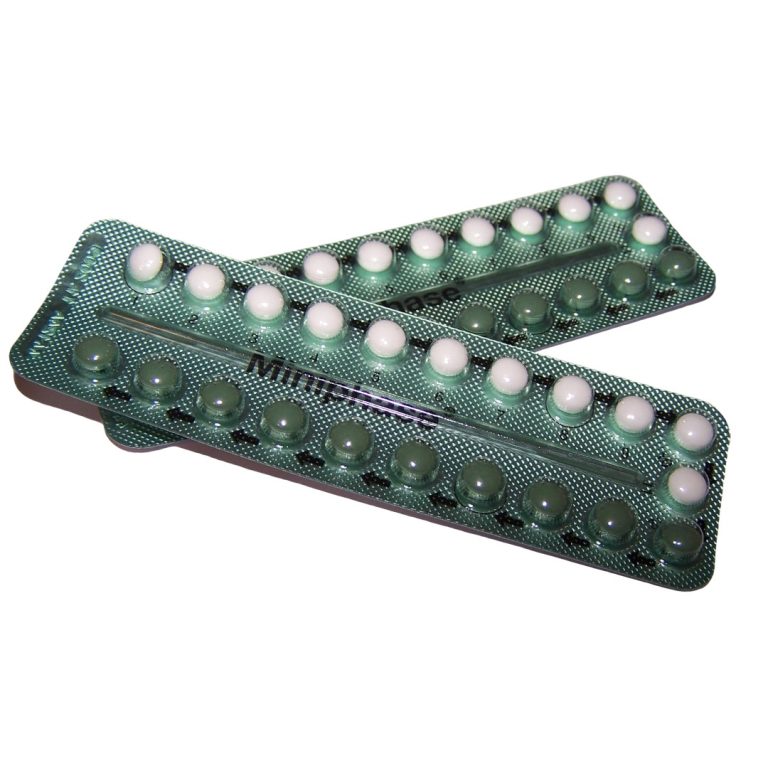
In 1970, the U.S. Food and Drug Administration (FDA) required the first patient package insert: oral contraceptives must contain…

On Dec. 13, 1969, Charles Edwards, M.D., becomes FDA commissioner.

On Dec. 2, 1969, the White House Conference on Food, Nutrition, and Health (WHC) was a seminal event…

On Sept. 4, 1969, the FDA issued a report that called birth control pills safe, despite a slight…

In 1969, the U.S. Food and Drug Administration (FDA) began administering Sanitation Programs for milk, shellfish, food service, and…
In 1969, live, attenuated rubella vaccines were first licensed in the U.S., and a vaccination program was established…
On Nov. 26, 1968, the U.S. Food and Drug Administration (FDA) licensed a second live, further attenuated measles…

On Apr. 7, 1968, the U.S. Food and Drug Administration (FDA) Bureau of Drug Abuse Control and Treasury Department…

In March 1968, a reorganization of federal health programs placed the U.S. Food and Drug Administration (FDA) in…

In 1968, the U.S. Food and Drug Administration (FDA) formed the Drug Efficacy Study Implementation (DESI) to implement recommendations…

In 1968, Medtronic annual sales skyrocketed to more than $12 million, with the company reporting net income in…
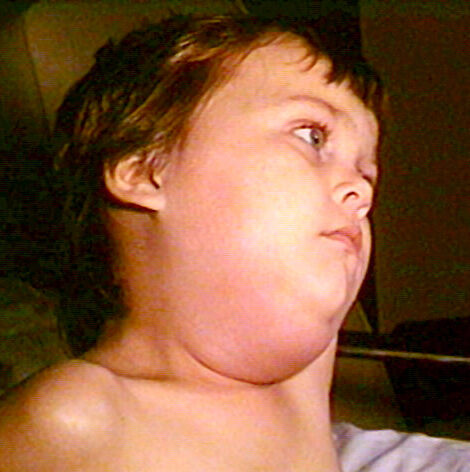
On Dec. 28, 1967, the U.S. Food and Drug Administration (FDA) approved Mercks mumps virus vaccine live (MumpsVax)….

In 1967 the Fair Packaging and Labeling Act (FPLA or Act), enacted by the U.S. Congress directs the…

In 1967, Medtronic introduced two “on-demand” pacemakers, designed to avoid competition between paced beats and the patient’s own…